Q1. When both positive and negative ions of a salt are complex ions and the two isomers differs in the distribution of ligands in the cation and the anion; it is a type of:
Solution
When both positive and negative ions of a salt are complex ions and the two isomers differs in the distribution of ligands in the cation and the anion; it is a type of Coordination isomerism.
Q2. What is meant by unidentate and bidentate ligands? Give example.
Solution
Ligand may contain one or more than one donor atoms. If only one donor atom is present in its molecule which can coordinate then it is unidentate. For example, H2O, NH3.
If the ligand contains two donor atoms placed in a way that a five or six membered ring is formed with the metal ion then it is called as bidentate. Example is oxalate ion.

Q3. Write step wise stability constant expressions K1 and K2 for the reaction Cu 2+ + 4NH3 ⇌ [Cu(NH3)4]2+
Solution
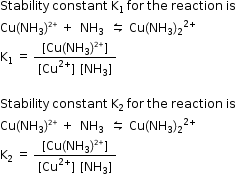
Q4. How will you distinguish between the two enantiomers of [PtCl2(en)2]+
Solution
Among the two enantiomers of [PtCl2(en)2]+, one isomer is dextrorotatory while the other is laevorotatory.

Q5. From the cis and trans isomer of [CrCl2(ox)2]3-which is chiral and optically active?
Solution
The two entities are represented as,
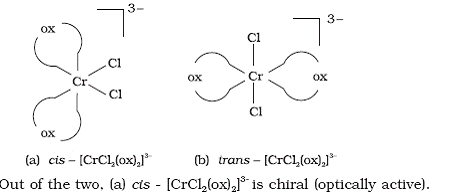 i.e. from the cis and trans isomer of [CrCl2(ox)2]3- optically active.
i.e. from the cis and trans isomer of [CrCl2(ox)2]3- optically active.
 i.e. from the cis and trans isomer of [CrCl2(ox)2]3- optically active.
i.e. from the cis and trans isomer of [CrCl2(ox)2]3- optically active.
Q6. What type of isomerism is exhibited by [Cr(H2O)5SCN]3+.
Solution
[Cr(H2O)5SCN]3+is a type of linkage isomer, as here more than one atom of a monodentate ligand may function as a donor. So the possible forms are [Cr(H2O)5SCN]3+ and [Cr(H2O)5NCS]2.
Linkage isomerism arises in a coordination compound containing ambidentate ligand.
Q7. How would you account for the following:[Ni(CO)4] has tetrahedral geometry while [Ni(CO)4]2- has a square planar geometry?
Solution
Nickel in [Ni(CO)4] is in the zero oxidation state and has electronic configuration of [Ar]3d84s2 or 3d10.sp3 hybrid orbitals accommodate four pair of electrons from four CO groups and the resulting tetrahedral complex is diamagnetic due to the absence of unpaired electrons.Nickel in [Ni(CO)4]2- is in the +2 oxidation state and has electronic configuration of [Ar]3d104s2. dsp2 hybrid orbitals accommodate four pair of electrons from four CO groups and the resulting square planer complex is diamagnetic due to the absence of unpaired electrons.
Q8. What is the IUPAC name of (NH4)3[Cr(SCN)6] complex?
Solution
IUPAC name of (NH4)3[Cr(SCN)6] is Ammonium hexathiocyanato-S-chromate(III).
Q9. What is ionisation isomerism? Give example.
Solution
Compounds which gives different ions in solution although they have same composition are called ionisation isomerism.
This form of isomerism arises when the counter ion in a complex salt is itself a potential ligand and can displace a ligand which can then become the counter ion.
An example is provided by the ionisation isomers [Co(NH3)5SO4]Br and [Co(NH3)5Br]SO4.
Another example is, [Pt(NH3)4(OH)2]SO4 and [Pt(NH3)4SO4](OH)2.
Q10. Draw the structure of [Cr(CO)6].
Solution
[Cr(CO)6] is octahedral in shape.

Q11. Write the formula of the co-ordination compound dichlorobis(ethylenediammine)cobalt(III) sulphate.
Solution
The formula of the co-ordination compound is [Co(en)2Cl2]SO4.
Q12. Draw the geometrical isomers of [Co(NH3)4Cl2]+.
Solution
Octahedral complex [Co(NH3)4Cl2]+exists as cis and trans isomer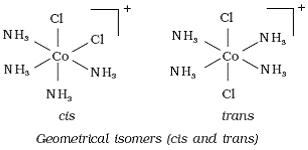

Q13. Explain the structure of [Co(NH3)6]3+.
Solution
The cobalt ion is in +3 oxidation state and has an electronic configuration of 3d6. Six pairs of electron one from each ammonia molecule occupy the six hybrid orbitals, hence the molecule is diamagnetic and octahedral in shape. 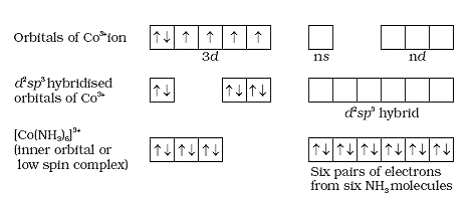

Q14. Which ligand has six donor atoms?
Solution
EDTA is a hexadentate ligand with six donor atoms, i.e, two nitrogen atoms and four oxygen atoms of the carboxylic acid groups capable of bonding to the metal atom.

Q15. Why [Cr(NH3)6]3+ is paramagnetic while [Ni(CN)4]2- is dimagnetic?
Solution
Chromium in [Cr(NH3)6]3+ is in +3 oxidation state and has electronic configuration of [Ar]3d54s1.
Cr3+ ion provides six empty orbitals to accommodate six pairs of electrons from six molecules of ammonia. The resulting complex involves d2sp3 hybridisation and is octahedral. Due to the presence of these unpaired electrons in the complex it is paramagnetic in nature.
Nickel in [Ni(CN)4]2- is in +2 oxidation state and has electronic configuration of [Ar]3d104s2.
dsp2 hybrid orbitals accommodate four pairs of electrons from four CN groups and the resulting square planner complex is diamagnetic due to the absence of unpaired electrons.

Q16. Write the structure of Zeises salt, Tetracarbonynickel and Ferrocene.
Solution
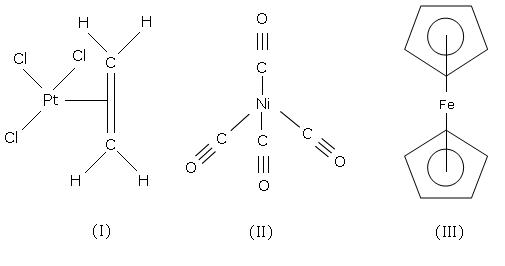 Zeisses salt, Tetracarbonynickel and Ferrocene
Zeisses salt, Tetracarbonynickel and Ferrocene
Comments
Post a Comment