Q1. Halogens have maximum negative electron gain enthalpy in their respective periods of the periodic table. Explain.
Solution
The halogens have a strong tendency to accept electrons due to their small size and effective nuclear charge so they have maximum negative electron gain enthalpy in their respective periods of the periodic table.
Q2. Write the products of hydrolysis of XeF6 with 1,2 and 3 moles of water.
Solution
The products of hydrolysis of XeF6 with 1,2 and 3 moles of water are:
XeF6 + H2O ------> XeOF4 + 2 HF
XeF6 + 2 H2O -----> XeO2F2 + 4 HF
XeF6 + 3 H2O -----> XeO3 + 6 HF
Q3. How SO2 is formed and discuss its state at room temperature. Why SO2 is a more powerful reducing agent in an alkaline medium than in an acidic medium.
Solution
Sulphur dioxide is formed together with a little (6-8%) sulphur trioxide when sulphur is burnt in air or oxygen.S(s) + O2(g) → SO2 (g)
It is a colourless gas and is highly soluble in water. It liquefies at room temperature under a pressure of two atmospheres.
The reducing action of SO2 is basically due to presence of HSO3- ion which contains one reducing H atom(S-H bond). In alkaline medium the concentration of HSO3- ion is more. So SO2 is a stronger reducing (H bond angle) agent in alkaline medium.
SO2 + H2O  HSO3- + H+
HSO3- + H+
Q4. Which oxoacids of phosphorus are reducing in nature?
Solution
All acids which have P-H bond are reducing in nature.

Q5. Why are halogens coloured?
Solution
Halogens are coloured because of their low ionisation energy.
As a result, the electrons get excited in the visible region of light.
The remaining light falls in the visible region and halogens appear to be coloured.
For example: Fluorine absorbs wavelengths corresponding to violet light and the remaining light appears to be yellow. Thus, fluorine has a yellow colour. Similarly, chlorine is greenish yellow, bromine reddish and iodine is a violet solid.
Q6. Mention the steps involved in the industrial preparation of nitric acid by Ostwalds process.
Solution
Nitric acid is produced industrially by the Ostwald process, which involves three steps:

Q7. Give reasons for the following: (i) Fluorine atom is more electronegative than iodine atom but HF has lower acidic strength than HI.(ii) The electron affinity of fluorine is less than that of chlorine but the oxidizing power of fluorine is greater than that of chlorine.
Solution
(i)
HF bond is very strong because of small size of fluorine atom. So the dissociation energy of HF is very high.
In case of HI, the dissociation energy of HI bond is low due to large size of iodine atom.
Hence, HI is a stronger acid than HF.
(ii) The reduction potential of fluorine is greater than that of chlorine. Therefore the electron affinity of fluorine is less than that of chlorine but the oxidizing power of fluorine is greater than that of chlorine.
Q8. Show that hydrogen peroxide behaves both as an oxidizing and reducing agent.
Solution
It produces oxygen and acts as an oxidizing agent in both acidic and basic medium.
As a oxidising agent : Mn2+ + H2O2→ Mn4+ + 2OH-
As a reducing agent: HOCl + H2O2→ H2O + Cl- + O2
Q9. PCl5 exists but NCl5 does not. Why?
Solution
Phosphorus can form PCl5 since it has vacant d-orbitals in its valence shell. NCl5 is not possible because of no vacant d-orbitals.
Phosphorus can form PCl5 since it has vacant d-orbitals in its valence shell. NCl5 is not possible because of no vacant d-orbitals.
Q10. Give reason H2S is a gas and H2O is liquid.
Solution
H2O is a liquid at room temperature because it is highly associated via intermolecular hydrogen bonding.
H2S cannot form hydrogen bonding due to large size of sulphur. So, H2S is a discrete molecule and the intermolecular forces of attraction are weak van der Waal’s forces.
Hence H2S is a gas at room tempearture.
Q11. Give the balanced reactions when chlorine reacts with: (i) Excess Ammonia (ii) Hot and conc. NaOH (iii) Hydrogen sulphide
Solution
(i)
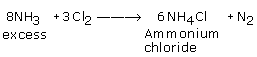 (ii) 6 NaOH + 3Cl2
(ii) 6 NaOH + 3Cl2  5NaCl + NaClO3 + 3H2O
(iii) H2S + Cl2
5NaCl + NaClO3 + 3H2O
(iii) H2S + Cl2  2HCl + S
2HCl + S
 (ii) 6 NaOH + 3Cl2
(ii) 6 NaOH + 3Cl2
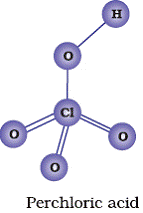
Q12. Draw the structures of following oxoacids of chlorine: (i) Chlorous acid (ii) Chloric acid (iii ) Perchloric acid
Solution
(i)
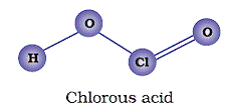 (ii)
(ii)
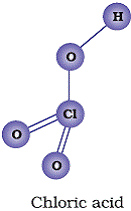 (iii)
(iii)
 (ii)
(ii)
 (iii)
(iii)

Q13. Why metals like chromium and aluminium do not react with powerful oxidizing agent like HNO3?
Solution
These elements form a passive layer of oxide on the surface and prevent the metal to react with nitric acid.
Q14. Write the steps involved in the manufacture of sulphuric acid by contact process.
Solution
Steps involved in the Contact process:
(1)Production of sulphur dioxide:
SO2 is produced by roasting metallic sulphides in air.
4FeS2 +11O2 → 2Fe2O3 + 8SO2
(2)Purification of gases:
To enhance the efficiency of catalyst, various impurities present in the mixture of sulphur dioxide and air are first removed.
(3)Catalytic oxidation of Sulphur dioxide:
Oxidation of SO2 to SO3 at 4500C in the presence of catalyst vanadium pentaoxide.
 (4) Absorption of sulphur trioxide in sulphuric acid:
Sulphur trioxide vapors are absorbed by stream of conc. sulphuric acid.
SO3 + H2SO4 → H2S2O7 (Oleum or pyrosulphuric acid)
(5)Dilution of oleum to obtain sulphuric acid:
Calculated amount of water is added to obtain sulphuric acid of desired strength.
H2S2O7 + H2O → 2H2SO4
(4) Absorption of sulphur trioxide in sulphuric acid:
Sulphur trioxide vapors are absorbed by stream of conc. sulphuric acid.
SO3 + H2SO4 → H2S2O7 (Oleum or pyrosulphuric acid)
(5)Dilution of oleum to obtain sulphuric acid:
Calculated amount of water is added to obtain sulphuric acid of desired strength.
H2S2O7 + H2O → 2H2SO4

Q15. What happens when sulphur is passed through conc. H2SO4 solution and SO2 is passed through an aqueous solution of Fe(III) salt?
Solution
Q16. How is oxygen prepared industrially and in the laboratory?
Solution

Q17. Why fluorine does not show variable valency while other halogens show variable valency?
Solution
Fluorine is the most electronegative element and cannot exhibit any positive oxidation state. Other halogens have d orbitals and therefore, can expand their octets and show + 1, + 3, + 5 and + 7 oxidation states also.
Q18. Draw the structure of XeOF4. How it is prepared?
Solution
XeOF4 is having a square pyramidal structure. It is prepared by treating XeF6 with H2O.
XeF6 + H2O ------> XeOF4 + 2 HF

Q19. The boiling point of NH3 is higher than the boiling point of PH3. Explain.
Solution
In NH3 the nitrogen atom is highly electronegative due to its small size so it undergoes hydrogen bonding and exists as an associated liquid where as PH3 molecule does not form hydrogen bonds due to the large size of phosphorus atom.
Q20. NH3 is soluble in water but PH3 is not soluble. Give reasons.
Solution
Ammonia forms hydrogen bonds but phosphorus does not form hydrogen bonds with water as a result ammonia does dissolve in water.
Q21. How is xenon difluoride prepared? What happens when it is treated with PF5?
Solution

Q22. Electron gain enthalpy of the elements of the halogen group becomes less negative down the group. But, why the negative electron gain enthalpy of fluorine is less than that of chlorine?
Solution
Due to small size of fluorine atom there are strong interelectronic repulsions in the relatively small 2p orbitals of fluorine atom and thus, the incoming electron does not experience much attraction. This is why the negative electron gain enthalpy of fluorine is less than that of chlorine.
Q23. Write the order of thermal stability of hydrides of group 16 elements.
Solution
On moving down the group, there is a decrease in the bond dissociation enthalpy(H-E) of hydrides with increase in atomic size.
So the thermal stability of hydrides decreases down the group.
H2O > H2S > H2Se>H2Te> H2Po
Q24. What is the chemical composition of bleaching powder?
Solution
The composition of bleaching powder is Ca(OCl)2.CaCl2.Ca(OH)2.2H2O.
Q25. Mention the conditions required to maximise the yield of ammonia.
Solution
(i) Moderate temperature 700K
(ii) High pressure 20MPa
(iii) Iron oxide + Al2O3 + K2O as catalyst
Q26. Why is phosphorus acid diprotic and phosphoric acid triprotic inpite 3 hydrogen atoms in both?
Solution

Q27. What is oleum? Draw its structure.
Solution
Oleum or pyrosulphuric acid(H2S2O7) is an oxoacid of sulphur.


Q28. Give chemical reactions involved in brown ring test to confirm nitrates.
Solution
The brown ring test depends on the ability of ferrous ion to reduce nitrates to nitric oxide, which reacts with ferrous ion to form a brown coloured complex.

Q29. Why noble gases have low boiling points?
Solution
Noble gases being monoatomic have no interatomic forces except weak dispersion forces and therefore, they are liquefied at very low temperatures. Hence, they have low boiling points.
Q30. Name two poisonous gases which can be prepared from chlorine gas.
Solution
(i) Phosgene (COCl2)
(ii) Tear gas (CCl3NO2)
Q31. Why does PCl3 fume in moisture?
Solution
It is a colourless oily liquid and hydrolyses in moisture giving fumes of HCl.
PCl3 + 3H2O → H2PO3 + 3HCl
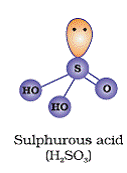
Q32. Draw the structure of Sulphurous acid.
Solution
Comments
Post a Comment