Q1. In the context of the Hall-Heroult process for the extraction of Al, which of the following statement is false?
Solution
Molten aluminium oxide (Al2O3) serves as an electrolyte in the extraction of aluminium.
Q2. Name the purest form of iron.
Solution
Wrought iron is the purest form of iron and contains carbon and other impurities less than 0.2%
Q3. What do you understand by the terms quenching and tempering. Also name the metal with which these terms are associated?
Solution
The hardness of steel is controlled by heat treatment. Following methods are used for this purpose: Quenching - In this process, the steel article is heated to redness (1123 K) and then suddenly cooled by plunging it in water or oil. The steel obtained by this treatment is hard and brittle. Tempering - In this process the quenched steel is heated to a temperature of about 550K and kept at that temperature for sometime and then cooled slowly, the steel obtained by this treatment is quite hard but less brittle.
Q4. Explain the process used for refining Zirconium or Titanium?
Solution
Van Arkel method is used for refining Zirconium or Titanium. In this method oxygen and nitrogen, present as impurity are removed by heating metal in an evacuated vessel with iodine. The metal iodide volatize and it is decomposed on a tungsten filament, heated to about 1800K. The pure metal is deposited on the filament.
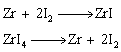
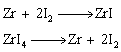
Q5. Describe the principle behind each of the following processes:
(i) Vapour phase refining of a metal.
(ii) Electrolytic refining of a metal.
(iii) Recovery of silver after silver ore was leached with NaCN.
Solution
(i) Vapour phase refining: It is the process of refining metal by converting it into its volatile compound and then, decomposing it to obtain a pure metal. The basic principle involved in this process is:
The metal should form a volatile compound with an available reagent, and the volatile compound should be easily decomposable so that the metal can be easily recovered.
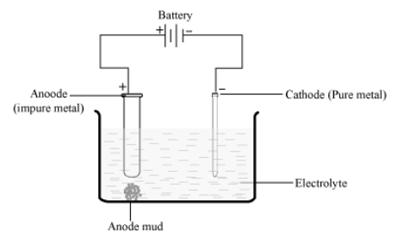
(ii) Electrolytic refining of a metal: It is the process of refining impure metals by using electricity. In this process, impure metal is made the anode and a strip of pure metal is made the cathode. A solution of a salt of the same metal is taken as the electrolyte. When an electric current is passed, metal ions from the electrolyte are deposited at the cathode as pure metal and the impure metal from the anode dissolves into the electrolyte in the form of ions. The impurities present in the impure metal gets collected below the anode. This is known as anode mud.

 (iii) In the process of leaching, the finely divided silver is treated with dilute solution of sodium cyanide while a current of air is continuously passed. As a result, silver pass into the solution forming solution dicyanoargenate(I) while the impurities remain unaffected which are filtered off.
(iii) In the process of leaching, the finely divided silver is treated with dilute solution of sodium cyanide while a current of air is continuously passed. As a result, silver pass into the solution forming solution dicyanoargenate(I) while the impurities remain unaffected which are filtered off.


 (iii) In the process of leaching, the finely divided silver is treated with dilute solution of sodium cyanide while a current of air is continuously passed. As a result, silver pass into the solution forming solution dicyanoargenate(I) while the impurities remain unaffected which are filtered off.
(iii) In the process of leaching, the finely divided silver is treated with dilute solution of sodium cyanide while a current of air is continuously passed. As a result, silver pass into the solution forming solution dicyanoargenate(I) while the impurities remain unaffected which are filtered off.

Q6. What is the significance of leaching in the extraction of aluminium?
Solution
Aluminium is extracted from bauxite which contains SiO2, iron oxide and titanium IV oxide as impurities. The ore is concentrated by digesting the powdered ore with 45% caustic soda solution at 473-523 K and 35-36 bar pressure. In this way Al2O3 is leached out as sodium aluminate.Al2O3.2H2O(s) + 2NaOH (aq) + H2O  2Na [Al (OH)4](aq)
The aluminate in solution is hydrolyzed by passing CO2 gas and hydrated Al2O3 is precipitated. Now the solution is seeded with freshly prepared sample of hydrated Al2O3 which induces precipitation. The sodium silicate remains in the solution and hydrated alumina is dried, filtered and heated to give back pure Al2O3.
2Na [Al (OH)4](aq)
The aluminate in solution is hydrolyzed by passing CO2 gas and hydrated Al2O3 is precipitated. Now the solution is seeded with freshly prepared sample of hydrated Al2O3 which induces precipitation. The sodium silicate remains in the solution and hydrated alumina is dried, filtered and heated to give back pure Al2O3.
Q7.
Describe how the following changes are brought about:
(i) Pig iron into steel.
(ii) Zinc oxide into metallic zinc.
(iii) Impure titanium into pure titanium.
OR
Describe the role of
(i) NaCN in the extraction of gold from gold ore.
(ii) SiO2 in the extraction of copper from copper matte.
(iii) Iodine in the refining of zirconium.
Write chemical equations for the involved reactions.
Solution
(i) Pig iron into steel:
Pig iron is converted to cast iron by heating molten pig iron with scrap iron and coke using hot air blast in specially designed furnaces. Cast iron is used in the manufacture of steel by mixing it with other metals.
(ii) Zinc oxide into metallic zinc:
Zinc oxide is converted to metallic zinc by reacting it with coke at 673 K.
 (iii) Impure titanium into pure titanium:
The crude titanium is heated in an evacuated vessel with iodine. The metal iodide being more covalent volatilizes.
(iii) Impure titanium into pure titanium:
The crude titanium is heated in an evacuated vessel with iodine. The metal iodide being more covalent volatilizes.
 Titanium iodide vapour are collected and then decomposed on a tungsten filament by electrically heating to about 1800K. The pure metal is thus deposited on the filament.
Titanium iodide vapour are collected and then decomposed on a tungsten filament by electrically heating to about 1800K. The pure metal is thus deposited on the filament.
 OR
(i) NaCN in the extraction of gold from gold ore:
In the metallurgy of gold, gold metal is leached with a dilute solution of NaCN in the presence of air (for O2). The gold metal is then obtained from the product by displacement reaction.
OR
(i) NaCN in the extraction of gold from gold ore:
In the metallurgy of gold, gold metal is leached with a dilute solution of NaCN in the presence of air (for O2). The gold metal is then obtained from the product by displacement reaction.
 (ii)SiO2 in the extraction of copper from copper matte:
Copper matte consists of Cu2S and FeS. In the converter FeS is converted to FeO. Silica helps in removal of FeO impurity as slag.
(ii)SiO2 in the extraction of copper from copper matte:
Copper matte consists of Cu2S and FeS. In the converter FeS is converted to FeO. Silica helps in removal of FeO impurity as slag.
 (iii) This method is very useful for removing all the oxygen and nitrogen present in the form of impurity in certain metals like Zr and Ti. The crude metal is heated in an evacuated vessel with iodine. The metal iodide being more covalent, volatilises:Zr + 2I2 → ZrI4 The metal iodide is decomposed on a tungsten filament, electrically heated to about 1800K. The pure metal is thus deposited on the filament.ZrI4 → Zr + 2I2
(iii) This method is very useful for removing all the oxygen and nitrogen present in the form of impurity in certain metals like Zr and Ti. The crude metal is heated in an evacuated vessel with iodine. The metal iodide being more covalent, volatilises:Zr + 2I2 → ZrI4 The metal iodide is decomposed on a tungsten filament, electrically heated to about 1800K. The pure metal is thus deposited on the filament.ZrI4 → Zr + 2I2
 (ii)SiO2 in the extraction of copper from copper matte:
Copper matte consists of Cu2S and FeS. In the converter FeS is converted to FeO. Silica helps in removal of FeO impurity as slag.
(ii)SiO2 in the extraction of copper from copper matte:
Copper matte consists of Cu2S and FeS. In the converter FeS is converted to FeO. Silica helps in removal of FeO impurity as slag.
 (iii) This method is very useful for removing all the oxygen and nitrogen present in the form of impurity in certain metals like Zr and Ti. The crude metal is heated in an evacuated vessel with iodine. The metal iodide being more covalent, volatilises:Zr + 2I2 → ZrI4 The metal iodide is decomposed on a tungsten filament, electrically heated to about 1800K. The pure metal is thus deposited on the filament.ZrI4 → Zr + 2I2
(iii) This method is very useful for removing all the oxygen and nitrogen present in the form of impurity in certain metals like Zr and Ti. The crude metal is heated in an evacuated vessel with iodine. The metal iodide being more covalent, volatilises:Zr + 2I2 → ZrI4 The metal iodide is decomposed on a tungsten filament, electrically heated to about 1800K. The pure metal is thus deposited on the filament.ZrI4 → Zr + 2I2
Q8. What is the role of each of the following in froth floatation process (i) Collectors (ii) Froth stabilizers (iii) Depressants
Solution
Collectors- They increase the non-wettability of mineral particles. Forth stabilizers- They are used to stabilize the froth formed. Depressants- They are used to prevent certain type of particles from forming the froth.
Q9. Name the various steps involved in metallurgy.
Solution
The various steps involved in metallurgy are: (i) Crushing and grinding of the ore (ii) Concentration of the ore (iii) Preliminary treatment of the concentrated ore (iv) Reduction (v) Purification or refining of crude metal
Q10. Describe Hall-Heroult process used for the electrolysis of pure alumina?
Solution
In Hall-Heroult process, purified Al2O3 is mixed with Na2AlF6 or CaF2 which lowers the melting point of matrix and brings conductivity. The fused matrix is electrolysed. The cathode and anode is made of steel and graphite respectively.
The various reactions which take place at cathode and anode are:

Q11. What is the difference between calcinations and roasting?
Solution

Q12. Define chromatography.
Solution
Chromatography is the process of separating the components of a mixture based on the difference in their adsorbability. The components are separated by mixing them with some suitable solvents. Different components are adsorbed at different levels on the chromatographic column.
Q13. Name the process by which Haematite and Magnetite ores can be concentrated. Also draw a well labeled diagram of this process.
Solution
Haematite and Magnetite ores can be concentrated by magnetic separation method.

Q14. Explain an extraction of a non-metal.
Solution
Extractions of non-metal such as chlorine is based on oxidation.
A very common example of extraction based on oxidation is the extraction of chlorine from brine (chlorine is abundant in sea water as common salt).
2Cl–(aq) + 2H2O(l) → 2OH–(aq) + H2(g) + Cl2(g) The ΔG0 for this reaction is + 422 kJ. When it is converted to E0 (using ΔG0 = – nE0F), we get E0 = – 2.2 V.
It will require an external e.m.f. that is greater than 2.2 V. But the electrolysis requires an excess potential to overcome some other hindering reactions. Thus, Cl2 is obtained by electrolysis giving out H2 and aqueous NaOH as byproducts.Electrolysis of molten NaCl is also carried out. But in that case, Na metal is produced and not NaOH.
Comments
Post a Comment