Q1. Explain preparation of sulphur sol and gold sol.
Solution
Sulphur sol and gold sol are Lyophobic sols and are unstable and are build to colloidal size from small particles by the Condensation method by Chemical reactions.
Oxidation sulphur sol is prepared by passing H2S into SO2
H2S + SO2 → S (sol) + H2O
Reduction of AuCl3 with formaldehyde gives gold sol.
AuCl3+ HCHO + 3H2O → Au (sol) + 3HCOOH + 6HCl
Q2. The relation between extent of adsorbtion (x/m) and pressure at constant temperature is called as:
Solution
Relation between extent of adsorbtion (x/m) and pressure at constant temperature is called isotherm.
Q3. Which of the following quantity/quantities have negative value during adsorption ?
Solution
As adsorption process is spontaneous where  G,
G,  S and
S and  H is negative.
H is negative.
 G,
G,  S and
S and  H is negative.
H is negative.
Q4. Formation of an ethyl ethanoate from ethanol and acetic acid in the presence of concentrated  is an example of:
is an example of:
 is an example of:
is an example of:Solution
Acid catalysed formation of an ester is an example of homogenous catalysis as reactant and catalyst have same phase.
Q5. The stabilisation of an emulsion by means of emulsifier is called:
Solution
The stabilisation of an emulsion by means of emulsifier is called as emulsification.
Q6. What are co-enzymes and activators?
Solution
Coenzymes are small non protein (vitamin) which enhances the activity of the enzyme considerably. Activators are generally metal ions. These metal ions, when weakly bonded to enzyme molecules, increase their catalytic activity. Amylase in presence of sodium chloride i.e., Na+ ions are catalytically very active.
Q7. How does a catalyst alter the rate of reaction?
Solution
Catalyst alters the rate of a reaction by the Adsorption and sorption. The probability of effective collision increases once the reactant is adsorbed on the surface and then desorps as product Catalyst forms a activated complex with the reactant which is at a lower energy level and provides an alternate path with lesser energy barrier.
Q8. What kind of colloid is fog?
Solution
Fog is an aerosol. Aerosols are colloidal solutions in which liquids are dispersed in a gas. In case of fog, tiny particles of water are suspended in the air. These occur naturally, as well as originating from volcanoes, dust storms, forest and grassland fires, living vegetation, and sea spray. Human activities, such as the burning of fossil fuels and the alteration of natural surface cover, also generate aerosols.
Q9. What are factors which influence adsorption of gas on solids?
Solution
The factors which influence adsorption of gas on solids are: (i) Nature of adsorbate (ii) Nature of adsorbent (iii) Temperature (iv) Pressure (v) Surface area exposed
Q10. Distinguish between Physisorption and Chemisorption on the basis of Bond, Layers and Temperature.
Solution
Property
Physisorption
Chemisorptions
Bond/ Interaction
Physical van der Waal's force between adsorbate and adsorbent e.g. gases on solids.
Chemical bond between adsorbate and adsorbent e.g. carbon monoxide on iron oxide.
Layers
Multi molecular layers
Mono molecular layers.
Temperature
Physisorption decreases with increase in temperature
Chemisorptions initially increases with increase in temperature as a chemical bond is formed, once the bond formation is complete it decreases with increase in temperature.
Q11. How is cream separated from milk which is an emulsion.
Solution
Suspended cream can be separated by centrifugation or ultracentrifugation.
Ultra centrifugation is a process in which rotation at high speed makes the colloid separates out from the medium.
Q12. What is the process involved when impurities are precipitated from water on addition of alum.
Solution
The process involved when impurities are precipitated from water on addition of alum is coagulation The role of alum in this process is that it coagulates particles present as impurities and deposits on the suspended clay particles in water. In this way water gets 'loaded' with alum and becomes heavy and settles down at the bottom.
Q13. Give a test to distinguish between two types of emulsions.
Solution
The following test can distinguish between Oil/Water and Water/Oil Dye test: On addition oil soluble dye to the colloid, background will be colored in Water/Oil emulsion and droplets will be colored in Oil/Water emulsion. DilutionTest: Diluted with water, water forms a separate layer in Oil/Water emulsion.
Q14. Define sorption.
Solution
When both the adsorption and absorption occur simultaneously and are not distinguishable, such a process is called sorption.
Q15. What is Brownian movement and why it is called so?
Solution
Browian movement is the continuous zig-zag motion of particles all over the field of view. when seen under ultra microscope. This motion was first observed by the British botanist, Robert Brown, and so known as Brownian movement
Q16. Write the differences between physisorption and chemisorption with respect to the following:
(i)Specificity
(ii)Temperature dependence
(iii)Reversibility and
(iv)Enthalpy change
Solution
S.NO
Point of difference
Physiosorption
Chemisorption
(i)
Specificity
It is not specific in nature.
It is highly specific in nature.
(ii)
Temperature
Dependence
Usually occurs at low temperature and decreases with increase in temperature.
Occurs at high temperature and increases with the increase in temperature.
(iii)
Reversibility
It is reversible in nature
It is irreversible in nature.
(iv)
Enthalpy change
Enthalpy of adsorption is low; of the order of 20-40 KJmol-1.
Enthalpy of adsorption is high; of the order of 80-240 KJmol-1.
Q17. Why is platinum used in combustion of hydrocarbons and the reactions that occur in the treatment of vehicle exhaust?
Solution
Platinum is especially good for reactions where selectivity is not so important, e.g. in the catalytic combustion of hydrocarbons and the reactions that occur in the treatment of vehicle exhaust.
Q18. What is a micelle? Explain formation of associate or aggregate colloid.
Solution
When molecules or ions that have both a hydrophobic and a hydrophilic end are dispersed in water, they associate, or aggregate, to form colloidal sized particle formed in water by the association of molecules or ions that each has a hydrophobic end and a hydrophilic end. The hydrophobic ends point inward towards one another and the hydrophilic ends are on the outside of the micelle facing the water. A colloid in which the dispersed phase consists of micelles is called an association colloid.
Example: Ordinary soap in water which consists of compounds such as sodium stearate, C17H35COONa. The stearate ion has a long hydrocarbon chain and hydrphilic end end because it is ionic.
 Soap and sodium lauryl sulphate (synthetic detergent present in toothpastes and shampoos):
CH3CH2CH2CH2CH2CH2CH2CH2CH2CH2CH2CH2OSO3−Na+
are examples of "anionic" molecules as they have a negative charge at the hydrophilic end. Other detergent molecules are "cationic", because they have a positive charge at the hydrophilic end:
Soap and sodium lauryl sulphate (synthetic detergent present in toothpastes and shampoos):
CH3CH2CH2CH2CH2CH2CH2CH2CH2CH2CH2CH2OSO3−Na+
are examples of "anionic" molecules as they have a negative charge at the hydrophilic end. Other detergent molecules are "cationic", because they have a positive charge at the hydrophilic end:
 Many cationic detergents also have germicidal properties and are used in hospital as disinfectants and in mouthwashes.
Many cationic detergents also have germicidal properties and are used in hospital as disinfectants and in mouthwashes.
Q19. What is peptization? Explain with example.
Solution
Peptization: Peptization may be defined as the process of converting a precipitate into colloidal sol by shaking it with dispersion medium with a small amount of electrolyte. The electrolyte used for this purpose is called peptizing agent. when ferric chloride is added to ferric hydroxide precipitate, the precipitate of hydroxide changes to sol by adsorbing ferric ions. Fe3+ is absorbed on Fe(OH)3 and breaks into colloids. 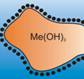 Black dots represent Fe3+
Black dots represent Fe3+
 Black dots represent Fe3+
Black dots represent Fe3+
Q20. What type of colloidal system foam is? Give example.
Solution
Foam is a dispersion of a gas in a liquid (liquid foams). Examples froth, whipped cream, soap lather.
Q21. The substances which are added to stabilise the emulsions are called:
Solution
The substances which are added to stabilise the emulsions are called emulsifying agent.
Q22. Explain how the phenomenon of adsorption finds applications in each of the following processes:
(i)Production of high vacuum
(ii)Heterogeneous catalysis
(iii)Froth floatation process
OR
(i)Micelles
(ii)Peptization
(iii)Desorption
Solution
(i)Production of high vacuum: Traces of air can be adsorbed by charcoal from a vessel, evacuated by a vacuum pump to give a very high vacuum.
(ii)Heterogeneous catalysis:The gaseous reactants are adsorbed on the surface of the solid catalysts. As a result, the concentration of the reactants increases on the surface and hence the rate of the reaction increases.
(iii)Froth floatation process: This process is used to remove gangue from sulphide ores. The basic principle involved in this process is adsorption.
In this process, a mixture of water pine oil is taken in tank. The impure powdered sulphide ore is dropped in through hopper and the compressed air is blown in through the agitator is rotator is rotated several times. As a result, froth is formed and the sulphide ores get adsorbed in the froth. The impurities settled down and are let out through an outlet at the bottom. The froth formed is collected in froth collector tank. After sometime, the ore particles in the froth collecting tank start settling gradually, which are then used for further metallurgical operations.
OR
(i)A micelle is an aggregate of surfactant molecules dispersed in a liquid. A micelle in aqueous solution forms as aggregate such that the hydrophilic "head" regions are in the centre of micelle.
(ii)Peptization is the process of conversion of a precipitate into a colloidal sol by shaking it with the dispersion medium in the presence of an electrolyte. The electrolyte used in this reaction is known as a peptizing agent.
(iii)Desorption is the process of removing an adsorbed substance from the surface through which it was adsorbed.
Q23. What is the difference between multimolecular and macromolecular colloids? Give one example of each type. How are associated colloids different from these two types of colloids?
Solution
Multimolecular colloids
Macromolecular colloids
On dissolution, a large number of atoms or smaller molecules of a substance aggregate together to form species having size in the colloidal range (diameter<1nm). The species thus formed are called multimolecular colloids.
Macromolecules in suitable solvents form solutions in which the size of the macromolecules may be in the colloidal range. Such systems are called macromolecular colloids.
Example: gold sol / sulphur sol
(Any one)
Example: starch, cellulose, proteins, enzymes, polythene, nylon, polystyrene, synthetic rubber (Any one)
Some substances at low concentrations behave as normal strong electrolytes, but at higher concentrations exhibit colloidal behaviour due to the formation of aggregates. The aggregated particles thus formed are called associated colloids or micelles. The formation of micelles takes place only above a particular temperature called Kraft temperature and above a particular concentration called critical micelle concentration. On dilution, these colloids revert back to individual ions.
Q24. Ferric hydroxide is a negative sol, which of the following electrolyte will coagulate it most:
Solution
1 mole FeCl3 will produce 3 negative charge, so has maximum coagulating power.
Q25. Explain how the phenomenon of adsorption finds applications in each of the following processes:
(i) Production of high vacuum
(ii) Heterogeneous catalysis
(iii) Froth floatation process
Or
Define each of the following terms:
(i) Micelles
(ii) Peptization
(iii) Desorption
Solution
(i) Production of high vacuum: Traces of air can be adsorbed by charcoal from a vessel, evacuated by a vacuum pump to give a very high vacuum.
(ii) Heterogeneous catalysis:The gaseous reactants are adsorbed on the surface of the solid catalysts. As a result, the concentration of the reactants increases on the surface and hence the rate of the reaction increases.
(iii) Froth floatation process: This process is used to remove gangue from sulphide ores. The basic principle involved in this process is adsorption.
In this process, a mixture of water pine oil is taken in tank. The impure powdered sulphide ore is dropped in through hopper and the compressed air is blown in through the agitator and rotator is rotated several times. As a result, froth is formed and the sulphide ores get adsorbed in the froth. The impurities settled down and are let out through an outlet at the bottom.
Or
(i) A micelle is an aggregate of surfactant molecules dispersed in a liquid. A micelle in aqueous solution forms an aggregate such that the hydrophilic "head" regions are in contact with the surrounding solvent and the hydrophobic tail regions are in the centre of micelle.
(ii) Peptization is the process of conversion of a precipitate into a colloidal sol by shaking it with the dispersion medium in the presence of an electrolyte. The electrolyte used in this reaction is known as a peptizing agent.
(iii) Desorption is the process of removing an adsorbed substance from the surface through which it was adsorbed.
Q26. What do you mean by emulsion and emulsification?
Solution
An emulsion is a dispersion of an insoluble liquid in another liquid. Process of preparing emulsions is called Emulsification.
Q27. Write short note on dispersion methods of preparation of colloids.
Solution
There are three dispersion methods:1. Mechanical dispersion - Suspension dimension are grinded in colloidal mill or ultrasonic disintegrator to colloidal dimensions.2. Electrical disintegration- Metal sols are prep by making electrode of the metal and immersed in dispersion medium and a electric arc is struck(involves dispersion plus condensation).3. Peptization- Precipitate is converted into colloids sol by shaking in dispersion medium with an electrolyte called peptizing agent. e.g. Fe(OH)3 ppt + FeCl3. Fe+++ is absorbed on Fe(OH)3 and breaks into colloids.
Q28. Explain mechanism of enzymatic reaction.
Solution
The mechanism is like a key fits into the lock and the active site of the enzyme fits into the substrate after which the enzyme separates and the substrate breaks into product. Steps1: Binding of enzyme to substrate to form activated complex. E + S  ES* Step2: Decomposition of activated complex to form product. ES*
ES* Step2: Decomposition of activated complex to form product. ES*  E + P
E + P
Q29. Describe some of the important features of catalysts.
Solution

Q30. (a) How can a colloidal solution and true solution of the same colour be distinguished from each other?
(b) List four applications of adsorption.
Or
Explain the following observation:
(a) Lyophilic colloid is more stable than lyophobic colloid.
(b) Coagulation takes place when sodium chloride solution is added to a colloidal solution of ferric hydroxide.
(c) Sky appears blue in colour.
Solution
(a) The path of light becomes visible when passed through the colloidal solution while it is not visible in case of true solution. This is because of Tyndall effect caused by the scattering of light by colloidal particles.
(b) Applications of adsorption:
(i) Activated charcoal is used in gas masks to remove poisonous gases such as CH4, CO, etc.
(ii) Animal charcoal is used as decolouriser in the manufacture of sugar.
(iii) Silica is used for removing moisture.
(iv) The ion exchange resins are used for removing hardness of water.
Or
(a) A lyophilic sol is stable due to the charge and the hydration of sol particles. Such a sol can only be coagulated by removing the water and adding solvents like alcohol, acetone, etc. and then an electrolyte.
On the other hand, a lyophobic sol is stable due to the charge only and hence can be easily coagulated by adding small amount of an electrolyte.
(b) The colloidal particles get precipitated i.e., ferric hydroxide is precipitated. When an excess of electrolyte (e.g., NaCl solution) is added to the colloidal solution of ferric hydroxide. This is because of the fact that colloids interact with the ions carrying the charge opposite (Cl-) to the one present on them. This causes neutralisation, which leads to their coagulation.
(c) The atmospheric particles of colloidal range scatter blue component of the white sunlight preferentially. That is why the sky appears blue.
Q31. State Hardy Schulz law.
Solution
Hardy Schulze law: Ions carrying opposite charge are effective in coagulation of solution and the coagulating power is directly proportional to the valency of the ion. For negative colloid (As2S3sol) the order of precipitation will be as follows Fe3+ > Ba2+ > Na+ ions. For positive colloid the order of precipitation will be as follows (Fe(OH)3) PO43-> SO42- > Cl-.
Q32. What is the difference between flocculation and coagulation?
Solution
Aggregation of colloidal particles into insoluble precipitate by addition of some suitable electrolyte is called coagulation or flocculation coagulation value: Minimum amount (in millimoles) of electrolyte required to cause complete coagulation in one litre colloidal solution in two hours is called precipitation or coagulation value.
Q33. Give reason why adsorbent is taken in finely divided state.
Solution
Solids particularly in finely divided state have large surface area and adsorb large quantity of the absorbate on its surface. So charcoal, silica gel, alumina gel, clay, colloids and metals in finely divided state are good adsorbent.
Q34. List conditions optimum for enzymatic reactions.
Solution
Conditions optimum for enzymatic reactions are: 1. Salt Concentration: More effective in dilute solutions. 2. Effects of Temperature: The optimum temperature for human enzymes is between 35 and 40oC. 3. Effects of pH: Most enzymes function between a pH of 6 and 8.
Q35. Why should colloids need purification?
Solution
If colloids are not separated from impurities the colloids will be unstable. Ionic impurities precipitate the colloid and so it is important to purify colloids.
Comments
Post a Comment