Q1. What does the expression  signify?
signify?


Solution
Average rate of reaction is the rate of reaction for a fixed interval of time.
Q2. In a reaction 2A + B → A2B, the reactant A disappears at:
Solution
According to the given equation, the rate of disappearance of reactant A is twice the rate of disappearance of reactant B.
Q3. The decomposition of N2O5 is first order reaction and represented by  After 15 minutes the volume of O2 produced is 9 cc and at the end of the reaction 35 cc. The rate constant is equal to
After 15 minutes the volume of O2 produced is 9 cc and at the end of the reaction 35 cc. The rate constant is equal to
Solution

Q4. The rate law for a reaction A + H2O → B is rate α [A]. Find the molecularity and order of the reaction.
Solution
The molecularity of the reaction is 2 and order of the reaction is 1.
Q5. What are the units for the rate constant of first order reaction?
Solution
The unit for the rate constant of first order reaction is s-1.
Q6. What are the two conditions that are necessary for effective collisions?
Solution
The two conditions that are necessary for effective collisions are:
i) The molecules should collide with sufficient kinetic energy also called the threshold energy.
ii) The molecules should have proper orientation.
Q7. Name the factors that influence the rate of a chemical reaction.
Solution
The rate of a chemical reaction is influenced by the following factors:
i) Concentration of the reactants
ii) Temperature of the reactants
iii) Nature of the reacting substances
iv) Presence of catalyst
v) Exposure to radiations
Q8. A foreign substance that increase the speed of a chemical reaction is called
Solution
A catalyst is a substance whose addition to a reaction enhances the rate.
Q9. For a reaction A → B, it is found that the rate of the reaction quadruples when the concentration of A is doubled. The rate for the reaction is Rate = k [A]x, where the value of x is:
Solution
Rate = k[A]x
When the rate of the reaction quadruples, Rate1 = 4 Rate
k[2A]x = 4 k[A]x
[2A]x/[A]x = 4
so, x=2
Q10. For the reaction
2A + B → 3C + D
Which of the following does not express the reaction rate?
Solution
For the reaction 2A + B → 3C + D,
The reaction rate is written as follows:
The reaction rate w.r.t. A  The reaction rate w.r.t. B
The reaction rate w.r.t. B  The reaction rate w.r.t. C
The reaction rate w.r.t. C  The reaction rate w.r.t. D
The reaction rate w.r.t. D  .
The products have a + sign because the concentration of the products increases with time.
Hence, the answer (1) is not the correct expression to represent the rate of reaction.
.
The products have a + sign because the concentration of the products increases with time.
Hence, the answer (1) is not the correct expression to represent the rate of reaction.
 The reaction rate w.r.t. B
The reaction rate w.r.t. B  The reaction rate w.r.t. C
The reaction rate w.r.t. C  The reaction rate w.r.t. D
The reaction rate w.r.t. D  .
The products have a + sign because the concentration of the products increases with time.
Hence, the answer (1) is not the correct expression to represent the rate of reaction.
.
The products have a + sign because the concentration of the products increases with time.
Hence, the answer (1) is not the correct expression to represent the rate of reaction.
Q11. For a chemical reaction 2A + B → C, the rate of formation of C is 0.25 mol L-1hr-1. What is the rate of disappearance of A and B?
Solution
According to the equation, the rate of disappearance of A is twice the rate of formation of C. So rate of disappearance of A = - 0.5 mol L-1 h-1 and that of B is -0.25 mol L-1hr -1
Q12. The instantaneous rate of reaction A  B is
B is  . What is the significance of plus and minus sign?
. What is the significance of plus and minus sign?
Solution
In the above equation the negative sign indicates the decrease in concentration of the reactant and a positive sign indicates the increase in concentration of the product.
Q13. Draw schematic graphs showing the following details of the first order reactions.i) Variation of the rate of reaction with change in the concentration of the reactant. ii) Variation of In [R] Vs t and Log [Ro]/[R] vs t.
Solution
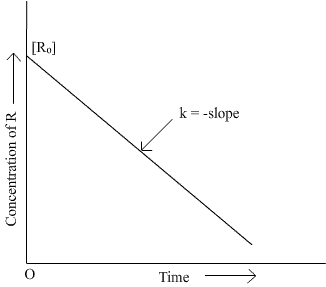
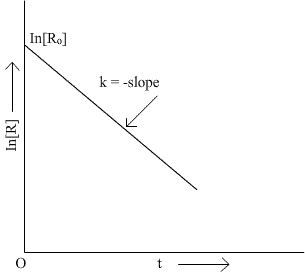
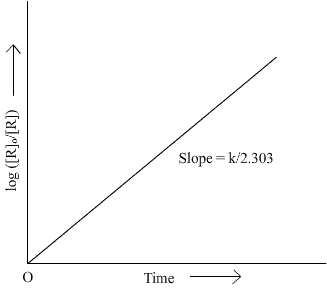
Q14. The rate constant of a first order reaction becomes 6 times when the temperature is increased from 350 K to 410 K. Calculate the energy of activation for the reaction.(R = 8.314 JK-1 mol-1)
Solution
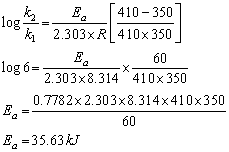
Q15. The rate of formation of nitric oxide (NO) in the following reaction is 3.6 x 10-3 mol L-1 s-1.
4NH3 (g) + 5O2 (g)  4NO2 (g) + 6H2O (g)
Find the rate of disappearance of Oxygen.
4NO2 (g) + 6H2O (g)
Find the rate of disappearance of Oxygen.
Solution
The rate of above reaction in terms of Oxygen and NO are
 Therefore rate of disappearance of Oxygen =
Therefore rate of disappearance of Oxygen = 

Q16. The experimental data for the reaction:
2A + B2 → 2AB is:
 Write the rate equation for the reaction?
Write the rate equation for the reaction?
Solution
When A is constant, the rate of the reaction doubles on doubling the concentration of B. i.e, Rate ∝ [Br2]1
When the concentration of B is constant, the rate of reaction remains same on doubling the concentration of A
i.e, rate ∝ [A]0.
Therefore, rate equation:

Q17. Identify A in the given diagram. .gif)
.gif)
Solution
Slope of the graph represents the instantaneous rate of reaction.
Q18. For a reaction the rate constants k1 and k2 calculated at two different temperatures T1=500 K and T2= 700 K are 0.025 s-1 and 0.07s-1 respectively. The values of A is:
Solution
Q19. The order of a reaction A + B  C is zero. Write its rate equation?
C is zero. Write its rate equation?
Solution
Rate = k [A] 0 [B] 0
Q20. The reaction, 2N2O (g) + O2 (g)  2NO2(g) proceeds through the following steps:
i) NO + O2
2NO2(g) proceeds through the following steps:
i) NO + O2  NO3 (fast)
ii) NO3 +NO
NO3 (fast)
ii) NO3 +NO  NO2 + NO3 (slow)
Predict the rate law expression for the reaction.
NO2 + NO3 (slow)
Predict the rate law expression for the reaction.
Solution
In the above reaction, the slowest step also called the rate determining step is
NO3 + NO  NO2 + NO3
Rate of the reaction = k [NO]2 [O2]
NO2 + NO3
Rate of the reaction = k [NO]2 [O2]
Q21. Differentiate between the order of a reaction and its molecularity.
Solution
Molecularity
Order of a reaction
i) It is the number of reacting species undergoing simultaneous collision in the reaction.
i) It is the sum of the powers of the concentration terms in the rate law expression.
ii) It is a theoretical concept.
ii) It is determined experimentally.
iii) It does not tell us anything about the mechanism of the reaction.
iii) It tells us about the slowest step and hence gives some clues about the mechanism of the reaction.
iv) It does not change with change in temperature and pressure.
iv) It changes with change in temperature
And pressure.
Q22. Calculate the activation energy of a hydrogen carbon whose decomposition is given as follows: K= (4.5 x 1011 s-1) e-28000K/T
Solution
According to Arrhenius equation k = A.e –Ea/RT
Comparing the given value of k = (4.5 x 1011 s-1) e-28000 K/T, we get,
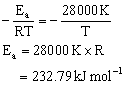
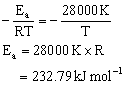
Q23. The half-life of a first order decomposition of nitramide is 2.1 hour at 25o C. Determine the time taken for the compound to decompose 99% of its original amount, rate constant = 0.2303 per hour.
Solution
Here a = 100
a – x = 100 – 99 = 1
t = 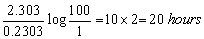
Q24. Which of the following statements is incorrect for the order of reaction:
Solution
The order of the reaction can be zero, whole number or fraction.
i.e, 0, 1, 2, 3 and a fraction.
Q25. The activation energies of two reactions are given as Ea1= 40 J and Ea2= 80 J, then the relation between their rate constants can be written as:
Solution
Use the relation  As the value of activation energy, Ea increases, the value of rate constant, k decreases.
So, k1 > k2 since E1 < E2
As the value of activation energy, Ea increases, the value of rate constant, k decreases.
So, k1 > k2 since E1 < E2
Q26. State the main postulates of the collision theory.
Solution
According to the collision theory:
i) A reaction occurs on collision of two molecules only if they possess a certain minimum amount of energy in excess of the normal energy of the molecules.
ii) The minimum energy which molecules must possess before collision should be equal to or greater than the activation energy.
Q27. The rate of reaction between A and B increases by a factor of 100. Calculate the order of the reaction when the concentration of A is increased 10 times.
Solution
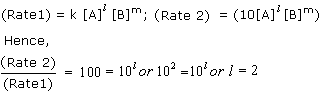
Q28. The activation energy for a reaction is zero. Calculate the value of its rate constant at 300 K, if k = 1.6 x 106 s-1 at 280 K? (R = 8.31 JK-1 Mol-1)
Solution
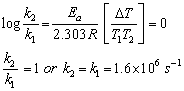
Q29. Define transition state or activation complex.
Solution
The arrangement of atoms corresponding to energy maxima (threshold energy) is called transition state or activated complex.
Q30. For a chemical reaction X  Y, The rate increases by the factor 2.25 when concentration of X is increased by 1.5. Derive or suggest the rate law equation and find the order of reaction.
Y, The rate increases by the factor 2.25 when concentration of X is increased by 1.5. Derive or suggest the rate law equation and find the order of reaction.
Solution
(Rate)1 = k[X]m2 (Rate)2 = k [1.5X]m Therefore, order of the reaction is 2. And rate law equation = k [X]2
Q31. The rate constant for the first order decomposition of certain reaction is described by the equation log k (sec-1) = 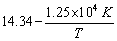 Calculate the activation energy for this reaction.
Calculate the activation energy for this reaction.
Solution
Given that log k = 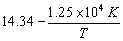 …(i)
According to Arrhenius equation
k = Ae-Ea/RT
…(i)
According to Arrhenius equation
k = Ae-Ea/RT
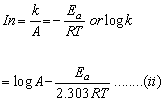 Comparing equation (i) and (ii), we have
Comparing equation (i) and (ii), we have
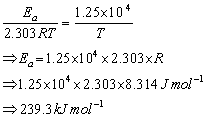
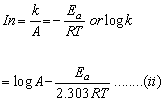 Comparing equation (i) and (ii), we have
Comparing equation (i) and (ii), we have
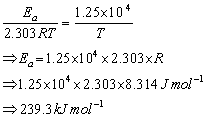
Q32. Define Collision frequency Z.
Solution
The number of collisions per second per unit volume of the reaction mixture is known as the collision frequency.

Comments
Post a Comment