Q1. Calculate the weight of copper deposited when two faraday of electricity is passed through a cupric salt. (Atomic weight of copper = 63.5)
Solution
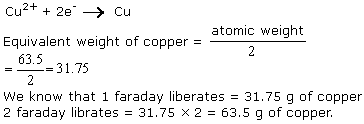
Q2. State Kohlrausch's Law of independent migration of ions.
Solution
Q3. Write the cell reactions which occur in lead storage battery (i) when the battery is in use (ii) when the battery is on charging.
Solution
At Anode : Pb (s) + SO4 2- (aq) → PbSO4 (s)+ 2e-.
At Cathode: PbO2(s) + SO4 2- (aq) + 4H+ (aq) +2e- →PbSO4 (s) + 2H2O(l)
The overall reaction is
Pb (s) + PbO2(s) + 4H+ (aq) + 2SO4 2-(aq) → 2PbSO4 (s) + 2H2O(l)
While charging:
At Cathode: PbSO4 (s)+ 2e- → Pb (s) + SO4 2- (aq)
At Anode: PbSO4 (s) + 2H2O(l) → PbO2(s) + SO4 2- (aq) + 4H+ (aq) +2e-
Q4. What is the function of a salt bridge in the electrochemical cell?
Solution
In the electrochemical cell a salt bridge performs the following functions:
a) It allows the flow of current by completing the circuit.
b) It maintains electrical neutrality.
Q5. Give three easier ways of preventing rusting of Iron.
Solution
Galvanization of Iron. By depositing a layer of zinc on the iron article the rusting of iron can be prevented.
By coating the surface of iron with a thin layer of paint or oil. They act as barrier and do not let the air and moisture come in contact with metal.
By connecting iron with magnesium rod or zinc rod as they are more reactive than Iron so they are preferentially corroded leaving Iron uncorroded.
Q6. Consider the following equations Au3+(aq) + 3e-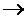 Au(s); EO = 1.50V AuCl4-+ 3e-
Au(s); EO = 1.50V AuCl4-+ 3e- 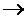 Au(s) + 2Cl-; Eo =1.00V i) Which of these species is the best oxidizing agent? ii) At which electrode would gold be deposited? iii) Out of AuCl3 and NaAuCl4, Au can be plated better from which solution?
Au(s) + 2Cl-; Eo =1.00V i) Which of these species is the best oxidizing agent? ii) At which electrode would gold be deposited? iii) Out of AuCl3 and NaAuCl4, Au can be plated better from which solution?
Solution
i) Au3+ is the best oxidizing agent.
ii) Gold would be deposited at the cathode.
iii) Gold can be plated better from AuCl3 (aq).
Q7. Calculate the reduction potential for the following half cell reaction at 298 K.
Ag+ (aq) + e- → Ag(s) given that (Ag+) = 0.1M and E° = + 0.80V
Solution
E = E0 - 0.0591/n log {1/Mn+}
E = 0.80V - 0.0591/1 log {1/0.1}
E = 0.80V - 0.0591/1 = 0.741V
Q8. How many coulombs of electricity are required for the oxidation of 90 g of water?
Solution
Q9. Write the Nernst equation and calculate the emf of the following cell at 298 K.Cu (s) | Cu2+ (0.130 M) || Ag+ (1.00M)| Ag (s) Given: EoCu2+/Cu = 0.34V and EoAg+/Ag = +0.80 V.
Solution

Q10. Can 1M ZnSO4 be stored in a vessel made up of copper?
Solution

Q11. Why each ion at infinite makes a unique contribution towards molar conductivity?
Solution
At infinite dilution or in extremely dilute solution the interionic interactions are the minimum and each ion moves independent of other. At this stage the ions moves freely and the electrostatic interaction between ions (attractive and repulsive) are minimized.
Q12. A lead storage cell acts both as Electrolytic cell as well as Galvanic cell. Explain.
Solution

Q13. Calculate the molar conductivity of a 0.8 M solution of electrolyte whose Resistivity is 5 ×10-3 ohm cm.
Solution
Here, the Resistivity = 5 ×10-3  cm
cm
 The conductivity of the solution κ =
The conductivity of the solution κ = 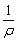 =
= 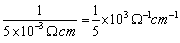 Molar conductivity
Molar conductivity 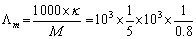 = 2.5 ×105
= 2.5 ×105 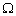 -1cm2mol-1.
-1cm2mol-1.
Q14. How does molar conductivity vary with concentration for strong and weak electrolytes? Explain?
Solution
The strong electrolytes are almost completely ionized in aqueous solution at all concentrations. So generally they have a high value of  m even at high concentrations. But, in concentrated solutions of strong electrolytes the density of ions is quite high which results in inter-ionic interactions. Due to these interactions, the value of
m even at high concentrations. But, in concentrated solutions of strong electrolytes the density of ions is quite high which results in inter-ionic interactions. Due to these interactions, the value of  m lowers at high concentrations. On increasing dilution the ions move apart which leads to the increase in the value of
m lowers at high concentrations. On increasing dilution the ions move apart which leads to the increase in the value of  m. The molar conductivity of a weak electrolyte increases with decrease in concentration.
m. The molar conductivity of a weak electrolyte increases with decrease in concentration.
 m even at high concentrations. But, in concentrated solutions of strong electrolytes the density of ions is quite high which results in inter-ionic interactions. Due to these interactions, the value of
m even at high concentrations. But, in concentrated solutions of strong electrolytes the density of ions is quite high which results in inter-ionic interactions. Due to these interactions, the value of  m lowers at high concentrations. On increasing dilution the ions move apart which leads to the increase in the value of
m lowers at high concentrations. On increasing dilution the ions move apart which leads to the increase in the value of  m. The molar conductivity of a weak electrolyte increases with decrease in concentration.
m. The molar conductivity of a weak electrolyte increases with decrease in concentration.
Q15. Iron does not rust even if the zinc metal coating on its surface is broken but the same is not true when coating is of Tin.
Solution
This can be explained from the position of these three elements in the electrochemical series. In comparison to Iron, Tin has lower reactivity than Iron where as Zinc is more reactive than Iron. When the coating of Zinc on the surface of Iron is broken even then Zinc will corrode in preference to Iron metal, whereas when coating of tin on Iron is cracked it exposes both the metals to external oxidizing conditions under this condition, Iron will oxidize in preference to Tin metal.
Q16. Calculate the reduction potential for the following half cell reaction at 298 K. Ag+(aq) + e- → Ag(s) Given that (Ag+) = )0.1M and E° = + 0.80V
Solution
EAg+|Ag = E0Ag+|Ag – ( 2.303 RT / n F ) log10 [Ag] / [Ag+]
= 0.80 – (0.059)/ 1 log10 1 / 0.1 = 0.80 – 0.059 = 0.741V
Q17. The products obtained from the electrolysis of molten NaCl and aqueous NaCl are different. Explain?
Solution
In the electrolysis of molten NaCl, sodium is deposited at the cathode while chlorine gas is liberated at anode. Where as in the electrolysis of aqueous solution of NaCl, Cl2 gas is liberated at anode and H2 gas is liberated at cathode.
Q18. Why a Dry cell is termed as dry Cell?
Solution
The Cell is filled with a moist electrolytic paste of manganese dioxide (MnO2) and charcoal and the second layer of ammonium chloride (NH4Cl) and zinc chloride (ZnCl2), the electrolyte is a dry electrolyte paste rather than solution, so it is called as Dry cell.
Q19. Why carbon dioxide dissolved in water increases the rate of rusting?
Solution
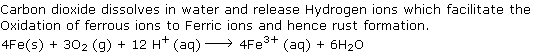
Q20. Account for the following: "Alkaline medium inhibits the rusting of iron".
Solution
In the process of rusting of Iron the hydroxide ions react with ferrous ions from the solution to give reddish brown substance ferrous hydroxide at the cathode. Hydroxide ions are formed by the reduction of Hydrogen ions, in alkaline medium the number of hydrogen ions decreases thereby decreasing the number of hydroxide ions in the solution , slowing the process of rusting of iron.
Q21. 

Solution
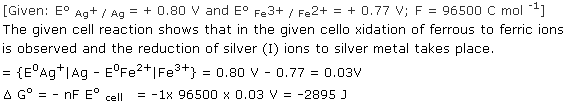
Q22. Name the reactions which take place at the cathode and anode during electrolysis?
Solution
During the electrolysis process, cathode involves reduction process and anode involves oxidation process at its surface.
Q23. Conductivity of 0.00250 M acetic acid is 8.00 x 10-5 S cm-1. Calculate its molar conductivity. If  0 for acetic acid is 300.0 S cm2 mol-1, what is its dissociation constant?
0 for acetic acid is 300.0 S cm2 mol-1, what is its dissociation constant?
 0 for acetic acid is 300.0 S cm2 mol-1, what is its dissociation constant?
0 for acetic acid is 300.0 S cm2 mol-1, what is its dissociation constant?Solution
( Λ m ) = k (conductivity) x 1000 / C (molar)
= (8.00 x 10-5 S cm-1 x 1000 cm3/L) / 0.00250 M
= 32.0 S cm2mol-1
α = 32.0/ 300 = 0.107
 K = 0.00250 x (32)2/ 300 ( 300-32)
= 0.00250 x 1024 / 300 x 268 = 0.00003184
K = 0.00250 x (32)2/ 300 ( 300-32)
= 0.00250 x 1024 / 300 x 268 = 0.00003184
 K = 0.00250 x (32)2/ 300 ( 300-32)
= 0.00250 x 1024 / 300 x 268 = 0.00003184
K = 0.00250 x (32)2/ 300 ( 300-32)
= 0.00250 x 1024 / 300 x 268 = 0.00003184
Q24. A solution of a salt of a metal of atomic weight 112 was electrolysed for 150 minutes with a current of 0.15 amperes. The weight of metal deposited was 0.783 mg; find the equivalent weight and valency of the metal in the salt.
Solution
Given, I = 0.15 ampere, t = 150 ×60 seconds
Q = I × t
Q = 0.15 ×150 × 60 coulombs = 1350 coulombs
Hence 1350 coulombs of electricity deposit = 0.783 gm of metal
96540 coulombs of electricity =
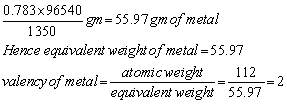
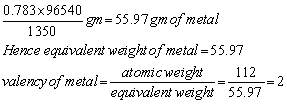
Q25. Calculate EMF of the following cell at 298 K. Ni(s) /Ni2+(1.0M) ll Ag+(1.0M)/Ag(s)
Solution
Given : E°Ni2+/Ni = - 0.25V and E°Ag+/Ag= -0.80V
E0 Cell = E°Ni2+/Ni - E° Ag+/Ag
= -0.25V-(-0.80) = 0.55V
Q26. Calculate the Λ0 for acetic acid, given, Λ0 (CH3COONa), Λ0 ( NaCl), Λ0 ( HCl) is 91 S cm-2mol-1, , 126 S cm-2mol-1 , 426 S cm-2mol-1 respectively. Calculate the degree of dissociation of the acid given the molar conductivity of the acid at the given concentration is 16.5 S cm-2mol-1. Λ0 ( CH3COOH) = ?
Solution
We know from Kohlrausch law that for
Λ0 ( CH3COONa) = λ0CH3COO- + λ0 Na + = 91S cm-2mol-1 ….(i)
Λ0 ( NaCl) = λ0Cl- + λ0 Na + = 126 S cm-2mol-1 …………. (ii)
Λ0 ( HCl) = λ0H + + λ0 Cl- = 426 S cm-2mol-1 ………….(iii)
Adding (i) and (iii) and subtracting (ii) from it we get,
Λ0 ( CH3COONa) + Λ0 ( HCl) = (λ0CH3COO- + λ0 Na +) + (λ0H + + λ0 Cl-)
Λ0 ( CH3COONa) + Λ0 ( HCl) = 91S cm-2mol-1 + 426 S cm-2mol-1 ………….(iv)
subtracting (ii) from (iv) we get,
(λ0CH3COO- + λ0 Na +) + (λ0H + + λ0 Cl-) - ( λ0Cl- + λ0 Na +) = λ0CH3COO- + λ0 H+
(λ0CH3COO- + λ0 Na +) + (λ0H + + λ0 Cl-) – ( λ0Cl- + λ0 Na +) = Λ0 ( CH3COOH)
Λ0 ( CH3COONa) + Λ0 ( HCl) - Λ0 ( NaCl) = 91S cm-2mol-1 + 426 S cm-2mol-1-126 S cm-2mol-1
Λ0 ( CH3COOH) = 517 S cm-2mol-1 - 126 S cm-2mol-1 = 391 S cm-2mol-1
Q27. What is the major application of Kohlrausch's Law.
Solution
This law enables us to calculate the molar conductivities of weak electrolytes. For weak electrolytes, Molar conductivity cannot be evaluated using graphical method. It can also be used to find out the dissociation constant of weak electrolytes.
Q28. What is the use of graphite rod in Lechlanche cell?
Solution
In Lechlanche cell a graphite rod is used as cathode.
Q29. Rusting of iron becomes quicker in saline medium. Explain.
Solution
Saline medium has extra salts such as sodium chloride dissolved in water. It has a greater concentration of electrolyte than ordinary medium. The ions present will favor the formation of more electrochemical cells and favor the transfer of hydrogen ions and will thus promote rusting or corrosion.
Q30. Why do we use inert electrolytes like KCl, KNO3, and NH4Cl in a salt bridge?
Solution
Inert electrolytes are used in a salt bridge because they do not chemically react with the solution in either of the compartment and they do not interfere with the net cell reaction also.
Q31. Calculate the molar conductivity of 1g of anhydrous barium chloride solution .Given that the conductivity of the solution is 0.0058S m-1and volume is 200cm3. (Atomic mass of Ba=137 and Cl= 35.5)
Solution
Conductivity ( ) = 0.0058 S cm-1
Molar mass of BaCl2 = 137 + 2 ×35.5 = 208 g mol-1
Number of moles of BaCl2 in 200 cm3 of solution =
) = 0.0058 S cm-1
Molar mass of BaCl2 = 137 + 2 ×35.5 = 208 g mol-1
Number of moles of BaCl2 in 200 cm3 of solution =  mol.
Volume of solution that contains 1 mol of BaCl2 (V) = 200 ×208cm3
Molar conductivity
mol.
Volume of solution that contains 1 mol of BaCl2 (V) = 200 ×208cm3
Molar conductivity  =
= 

Q32. Explain the significance of the negative value of electrode potential of an electrode taking example of magnesium metal.
Solution
Let us compare the Eo value for the magnesium metal and Hydrogen ions.
 Eo = -2.37V
2H+ (aq) + 2e-
Eo = -2.37V
2H+ (aq) + 2e-  H2(g) Eo = 0.00V
H2(g) Eo = 0.00V
 We know that for Magnesium electrode the standard electrode potential is negative.
It indicates that Conversion of Magnesium ions to magnesium metal is not favored.
However the reverse Conversion of the metal atoms to metal ions is more favored.
We know that for Magnesium electrode the standard electrode potential is negative.
It indicates that Conversion of Magnesium ions to magnesium metal is not favored.
However the reverse Conversion of the metal atoms to metal ions is more favored.
 Eo = -2.37V
Eo = -2.37V
 We know that for Magnesium electrode the standard electrode potential is negative.
It indicates that Conversion of Magnesium ions to magnesium metal is not favored.
However the reverse Conversion of the metal atoms to metal ions is more favored.
We know that for Magnesium electrode the standard electrode potential is negative.
It indicates that Conversion of Magnesium ions to magnesium metal is not favored.
However the reverse Conversion of the metal atoms to metal ions is more favored.
Q33. Write down the reactions taking place at the electrodes of a Mercury cell.
At Anode: The Oxidation of Zinc metal takes place and the Zinc ions react with the OH- ions from the electrolyte to form Zinc hydroxide.
Solution
Zn (s) → Zn2+ 2e-
Zn2+ + 2 OH- → Zn (OH)2
At Cathode: The reduction of Mercuric Oxide to mercury metal takes place.
HgO (s) + H2O + 2e- → Hg (l) + 2OH-
The Overall reaction is :
Zn (s) + HgO (s) →Zn (OH)2 + Hg (l)
Q34. On which factors does electrical conductivity of the solution of electrolyte depend?
Solution
The electrical conductivity of solution depends on the following factors: (A) The inter-ionic attraction: Larger inter- ionic attraction lesser is freedom of movement and smaller will be the electrical conductivity. (B) The salvation of ions: Larger the solute-solvent interaction greater is the extent of salvation and lower will be the electrical conductivity of the solution. (C) Viscosity of solvent: Larger the viscosity more will be the hindrances to the movement of ions. Lower will be the electrical conductivity. (D) Temperature: Increase in temperature leads to increase in kinetic energy which automatically increases the conductance of electrolytic solutions.
Comments
Post a Comment