Q1. Why Carbon tetrachloride and water are immiscible?
Solution
There is also a significant difference between the solvents: CCl4 and H2O. The difference between the electro negativities of the carbon and chlorine atoms in CCl4 is so small (EN = 0.56) that there is relatively little ionic character in the C Cl bonds.
Even if there was some separation of charge in these bonds, the CCl4 molecule wouldn't be polar, because it has a symmetrical shape in which the four chlorine atoms point toward the corners of a tetrahedron, as shown in the figure below. CCl4 is therefore best described as a nonpolar solvent.
Cl bonds.
Even if there was some separation of charge in these bonds, the CCl4 molecule wouldn't be polar, because it has a symmetrical shape in which the four chlorine atoms point toward the corners of a tetrahedron, as shown in the figure below. CCl4 is therefore best described as a nonpolar solvent.
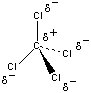 The difference between the electronegativities of the hydrogen and oxygen atoms in water is much larger (EN = 1.24), and the H
The difference between the electronegativities of the hydrogen and oxygen atoms in water is much larger (EN = 1.24), and the H O bonds in this molecule are therefore polar. If the H2O molecule was linear, the polarity of the two O
O bonds in this molecule are therefore polar. If the H2O molecule was linear, the polarity of the two O H bonds would cancel, and the molecule would have no net dipole moment. Water molecules are not linear, however, they have a bent, or angular shape. As a result, water molecules have distinct positive and negative poles, and water is a polar molecule, as shown in the figure below. Water is therefore classified as a polar solvent.
H bonds would cancel, and the molecule would have no net dipole moment. Water molecules are not linear, however, they have a bent, or angular shape. As a result, water molecules have distinct positive and negative poles, and water is a polar molecule, as shown in the figure below. Water is therefore classified as a polar solvent.
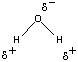 Because water molecules are bent, or angular, they have distinct negative and positive poles. H2O is therefore an example of a polar solvent
So because of their different nature the two do not dissolve with each other and on mixing them we see two separate layers of two solvents.
Because water molecules are bent, or angular, they have distinct negative and positive poles. H2O is therefore an example of a polar solvent
So because of their different nature the two do not dissolve with each other and on mixing them we see two separate layers of two solvents.
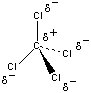 The difference between the electronegativities of the hydrogen and oxygen atoms in water is much larger (EN = 1.24), and the H
The difference between the electronegativities of the hydrogen and oxygen atoms in water is much larger (EN = 1.24), and the H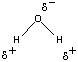 Because water molecules are bent, or angular, they have distinct negative and positive poles. H2O is therefore an example of a polar solvent
So because of their different nature the two do not dissolve with each other and on mixing them we see two separate layers of two solvents.
Because water molecules are bent, or angular, they have distinct negative and positive poles. H2O is therefore an example of a polar solvent
So because of their different nature the two do not dissolve with each other and on mixing them we see two separate layers of two solvents.
Q2. A solution containing 10.2g of glycerin per litre is found to be isotonic with 2% solution of glucose (molar mass = 180 g mol-1). Calculate the molar mass of glycerin.
Solution
For solution of Glucose pA V= n RT ----- (1)
pA x 0.1L = ( 2/180 )x R x T
For solution of Glycerin pB V= n RT ----- (2)
pB x 1L = ( 10.2/MB )x R x T
pA = pB
( 2/180 )x R x T/ 0.1 = ( 10.2/MB )x R x T/ 1
MB = 91.8 gmol-1
Q3. The freezing depression of 0.1M sodium chloride solution is nearly twice that of 0.1 M glucose solution. Explain?
Solution
Sodium chloride being ionic compound ionizes as (NaCl  Na++ Cl-) in aqueous solution. The concentration of solute particles in this case becomes approximately 0.2 M which is twice the concentration of glucose solution. Consequently, freezing point depression of NaCl solution is also approximately twice that of glucose solution.
Na++ Cl-) in aqueous solution. The concentration of solute particles in this case becomes approximately 0.2 M which is twice the concentration of glucose solution. Consequently, freezing point depression of NaCl solution is also approximately twice that of glucose solution.
Q4. Calculate the amount of KCl must be added to one kilogram of water so as to reduce its freezing point by two Kelvin. For water Kf = 1.86 K Kg mol-1.
Solution
ΔTf = i Kf m
ΔTf = i Kf no of moles / mass of solvent
ΔTf = 2 x 1.86 x n /74.5
n= 2 x 74.5 / 2 x 1.86 = 40.05g.
Q5. How much glucose, C6H12O6 must be dissolved per litre of the solution that yields osmotic pressure of 2.72 atmospheres at 300 K? R = 0.08221 L atm K-1 mol-1
Solution
p V= nRT
2.72atm x 1L = n x 0.082 x 300
n x 0.082 x 300 = 2.72atm x 1L
n = 2.72 atm x 1L/ 0.082 x 300
n = 0.110
mass of solute = 0.110 x 180= 19.90g
Q6. How many grams of HCl are in0 .075 L of 8M HNO3 solution (wt of HCl = 36.5 g/mol-1)?
Solution
8M solution means 8 moles of HCl are present in 1L solution so in .075Lof solution amount of HCl = 0.60 mol, 1mole weighs = 36.5g therefore weight of = 36.5g x .60 = 21.9g
Q7. What is negative deviation from ideal solution .Give an example?
Solution
A negative deviation from Raoult's law means that the vapor pressure of solution is lower than expected vapor pressure above the solution. If the solute A-A bond and solvent B-B is weaker than A-B bond i.e. solute and solvent are tightly bound to each other as they are to themselves, then the solution will show a negative deviation from Raoult's law because the solvent molecules will not find it easier to escape from solution into the gas phase.Acetone and chloroform form non ideal solution showing negative deviation from Raoult's law, hydrogen bond is created between acetone and chloroform as a result escaping is lesser then expected by Raoult's law and so it will show negative deviation.

Q8. Osmotic pressure of a solution containing 7g of dissolved protein per 100 cm3 of solution is 25mm Hg at temperature of 300K. Calculate the molecular mass of protein (R = 0.0821 L atmK-1mol-1).
Solution
 p = 25/760 =0.033atm
MB = 7g x 0.082 x 300/ 0.033 x 0.1L
MB = 52181gmol-1
p = 25/760 =0.033atm
MB = 7g x 0.082 x 300/ 0.033 x 0.1L
MB = 52181gmol-1
Q9. Calculate the amount of NaCl must be added to 1000 ml of water so as to reduce its freezing point by two Kelvin. For water Kf = 1.86 K Kg mol-1 , give that the density of water is 1.0 g ml-1 and NaCl is completely dissociated.
Solution
Mass of water = density x volume= 1 x 1000 = 1000g = 1Kg.
ΔTf = i Kf m
2 = (2 x 1.86 x z / 58.5 x 1000) x 1000
z = 58.5/1.86= 31.45g
Q10. The boiling point of a solution of 0.105 g of a substance in 15.84 g of ether was found to be 0.1000C higher than that of pure ether. What is the molecular mass of the substance? (Molal elevation constant for ether per 100g is 21.6)
Solution
Q11. Osmotic pressure is generally preferred for the determining the molecular masses of proteins. Explain.
Solution
Other methods of determining molar mass such as depression in freezing point, elevation in Boiling point are not useful because the magnitude of these Colligative properties for solution is very small. Moreover at higher temperature the proteins tend to denature.
Q12. Why for Sodium sulphate the solubility shows decrease on increasing temperature of solution.
Solution
The solubility of solutes is dependent on temperature. When a solid dissolves in a liquid, a change in the physical state of the solid analogous to melting takes place. Heat is required to break the bonds holding the molecules in the solid together. At the same time, heat is given off during the formation of new solute -- solvent bonds.
If the heat given off in the dissolving process is greater than the heat required to break apart the solid, the net dissolving reaction is exothermic (energy given off). On increasing the temperature as in accordance with the Le chateliers principle the equilibrium shifts in the backward direction favoring recrystallisation of salt.
Q13. The depression in freezing point is a colligative property. Explain.
Solution
The freezing point depression depends upon the molal concentration of the solute and does not depend upon the nature of the solute .It is therefore, a colligative property.
Q14. The vapor pressure of water at 293K is 23.75 mm Hg. A solution of substance P is prepared by dissolving 5 g of sugar in 100 g of water. The vapor pressure of resultant solution is 23.375mm Hg. Calculate the molecular mass of P.
Solution
[Po - Ps]/ Po = relative lowering of vapor pressure =x2
[Po - Ps]/ Po = n2/ n1 + n2
n2 = mass of P/ molar mass of P……….
n2 = 5.0 /MP
n2 = mass of water/ molar mass of water……….
n2 = 100/18= 5.50
[Po - Ps]/ Po = (5/ MP) / 5.50
[23.75 – 23.375]/ 23.375 =(5/ MP) / 5.50
0.0160 =(5/ MP) / 5.50
x = 56.8 g mol-1
Q15. 0.02molal solution of acetic acid is 3% dissociated at 25oC calculate the Osmotic pressure of the solution.
Solution
p = i CRT
Calculation of Van't Hoff factor
CH3COOH CH3COO- + H+
1 0 0
1-α α α
i = 1+ α /1 = 1+ 0.03 / 1= 1.003
p = i CRT
p = 1.003 x 0.0821 x 300 x 0.02 = 0.494atm
Q16. A 5% aqueous solution of cane sugar has a freezing point of 271K. Calculate freezing point of 5 % aqueous solution of glucose. The freezing point of pure water is 273.15K.
Solution
Q17. Two molar solution of potassium Ferro cyanide has degree of dissociation 0.70 at 300K.Find out the osmotic pressure of the solution.
Solution
p = i CRT
Calculation of Van't Hoff factor
K3[ Fe(CN) 6] 3 K+ + [ Fe(CN) 6]3-
1 0 0
1-α 3α α
i = 1+ 3α /1 = 1+ 3x0.70 / 1= 3.10
p= i CRT
p = 3.10 x 2 x 0.0821 x 300= 152.70 atm
Q18. Why molality is more preferred way of measuring strength than molarity.
Solution
Molarity changes with temperature as the volume will vary with increase or decrease in temperature but molality remains the same.
Q19. Define Azeotropes? Give an example.
Solution
The liquid mixtures which boil at constant temperature and can distill over as if a pure liquid is called azeotrope. Azeotrope behaves like a pure liquid and both of its components distill together. Ethanol and water forms an azeotrope mixture boiling at 78.15oC. The composition of the mixture is (95.5% ethyl alcohol and 4.5% water).
Q20. Vapour pressure of two liquid A and B are 120 and 180mm Hg at a given temperature. If 2 mole of A and 3 mole of B are mixed to form an ideal solution, calculate the vapour pressure of solution at the same temperature.
Solution
As per Raoult’s law Ptotal = X1 P1o + X2 P2o
P1= partial pressure of component one and P2= partial pressure of component two
Mole fraction of component one= X1 and Mole fraction of component two= X2
P1o and P2o are vapor pressure of a pure components one and two respectively.
X1 = 2/ 2+3 = 0.4 X2 = 1-0.4=0.6
Ptotal = 120 x 0.4 + 180 x 0.6 = 48.0+ 84.0 = 132 mmHg.
Q21. Equimolar solution of glucose and Common salt are not isotonic. Why?
Solution
Glucose is a non electrolyte, when added to water it do not break up into ions whereas Common salt is an electrolyte when added to water it breaks up to give Sodium and chloride ions , The number of particles in solution of Common salt are nearly double the number of particles in the solution of glucose so the osmotic pressure of common salt solution is nearly twice that if Glucose solution.
Q22. What is molal elevation constant? What are its units?
Solution
The molal elevation constant of the solvent is defined as the elevation in its boiling point when one mole of non-volatile solute is dissolved per kilogram (1000g) of solvent. Its unit is K.kg mol-1.
Comments
Post a Comment