Q1. Which among the following is a non-reducing sugar?
Solution
Sucrose is a non-reducing sugar because anomeric carbon of both the monosaccharides are involved in glycoside or acetal formation. So it does not have free -CHO group.
Q2. Despite having an aldehydic group:(a) Glucose does not give 2, 4-DNP test. What does this indicate?(b) Draw the howorth structure of  -D-(+)-Glucopyranose.(c) What is the significance of D and (+) here?
-D-(+)-Glucopyranose.(c) What is the significance of D and (+) here?
Solution
(a) This indicates that the aldehydic group in glucose is not free. (b) (c) 'D' gives the configuration, i.e. the group at carbon 5 is on the right hand side and (+) indicates that the isomer is Dextrorotatory.
(c) 'D' gives the configuration, i.e. the group at carbon 5 is on the right hand side and (+) indicates that the isomer is Dextrorotatory.
 (c) 'D' gives the configuration, i.e. the group at carbon 5 is on the right hand side and (+) indicates that the isomer is Dextrorotatory.
(c) 'D' gives the configuration, i.e. the group at carbon 5 is on the right hand side and (+) indicates that the isomer is Dextrorotatory.
Q3. What are reducing and non reducing sugars? What are the structural feature characterizing reducing sugars?
Solution
Sugars which can reduce Tollen's reagent or Fehling's solution are called reducing sugars. The sugars which don't reduce Tollen's reagent or Fehling's solution are called non-reducing sugars.The characteristics structural features of reducing sugars is the presence of either aldehydic group; (-CHO) such as glucose, mannose, galactose, etc. or α-ketol group (-CO-CH2OH) as present in fructose.
Q4. What type of bonding is present in Globular proteins?
Solution
Globular proteins involves proteins are hydrogen bonds, disulphide linkages, van der Waals and electrostatic forces of attraction.
Q5. How a nucleoside differs from nucleotide?
Solution
Both nucleotide and nucleoside have pentose sugar and nitrogenous base. Nucleotide differs from nucleoside with the presence of phosphoric acid whereas nucleoside do not have phosphoric acid.
Q6. Choose the correct option of a fat soluble vitamin from the following:
Solution
Vitamin A, D, E and K are fat soluble vitamins. Whereas, vitamin B and C are water soluble.
Q7. What are the components of nucleic acid?
Solution
Nucleic acids are biopolymers and the repeating unit in a nucleic acid is a nucleotide. All nucleotides on hydrolysis gives a mixture of three different types of compounds. These are sugar molecule, heterocyclic nitrogenous bases and phosphoric acid.
Q8. Give one example each for reducing and non-reducing sugars.
Solution
Reducing sugar: D - fructoseNon-reducing sugar: Sucrose
Q9. What are the Pyrimidine bases in DNA?
Solution
Pyrimidine bases in DNA are the Cytosine and Thymine.
Q10. Write any three difference between DNA and RNA.
Solution
DNA (Deoxyribonucleic acid)
RNA (Ribonucleic acid)
(i) DNA contains Cytosine and Thymine as pyrimidine bases and Guanine and Adenine as purine bases.
(i) RNA contains Cytosine and Uracil as pyrimidine bases and Guanine and Adenine as purine bases.
(ii) DNA has double stranded alpha-helix structure.
(ii) RNA has single stranded structure.
(iii) DNA occurs in nucleus of the cell.
(iii) RNA occurs in cytoplasm of the cell.
(iv) The sugar present in DNA is 2-deoxy-D(-) ribose.
(iv) The sugar present in RNA is D(-) ribose.
Q11. What type of bonding occurs in β-plated structure of proteins?
Solution
In β-structure, all peptide chains are stretched out to nearly maximum extension and then laid side by side which are held together by intermolecular hydrogen bonds between  and -NH group. The structure resembles the pleated folds of drapery and therefore is known as β-pleated sheet.
and -NH group. The structure resembles the pleated folds of drapery and therefore is known as β-pleated sheet.
Q12. Glucose reacts with acetic anhydride to form:
Solution
Glucose react with acetic anhydride to form penta-acetate as a molecule of glucose contains five -OH groups.

Q13. What are essential and non-essential amino acids? Give two examples of each type.
Solution
The amino acids, which can be synthesized in the body, are known as nonessential amino acids. For example - Glycerine and AlanineThe amino acids which cannot be synthesized in the body and must be obtained through diet, are known as essential amino acids. For example - Valine and Leucine
Q14. Describe what do you understand by primary structure and secondary structure of proteins.
Solution
(i) Primary structure of proteins:Each polypeptide chain in a protein has amino acids linked with each other in a specific sequence. This sequence of amino acids is said to be the primary structure of proteins.
(ii) Secondary structure of proteins: The secondary structure of proteins refers to the shape in which a long polypeptide can exist. The two different secondary structures possible are α-Helix structure and β-pleated sheet structure.
α-Helix structure: In α-Helix structure, a polypeptide chain forms all possible hydrogen bonds by twisting into a helix with -NH group of each amino acid residue and hydrogen bonded to >C=O of an adjacent turn of helix.
β-Helix structure: In a β-pleated structure, all peptide chains are stretched out of nearly maximum extensions and then laid side by side which are held together by intermolecular hydrogen bonds.
Q15. Why is Glucose given to patients under exhaustion?
Solution
Glucose is an instant source of energy and hence is given to patients under exhaustion.
Q16. What is essentially the difference between α-form of glucose and β-form of glucose? Explain.
Solution
The α-form of glucose and β-form of glucose can be distinguished by the position of the hydroxyl group on the first carbon atom.
In open chain α-glucose, the hydroxyl group on the first carbon atom is towards the right whereas, in the closed ring α- glucose, the hydroxyl group on the first carbon atom is below the plane of the ring.
On the other hand, in open chain β-glucose, α-glucose, the hydroxyl group on the first carbon atom is towards the left whereas, in the closed ring α-glucose, the hydroxyl group on the first time atom is above the plane of the ring.
The structures of open and closed α-form and β- form of glucose can be drawn as follows.
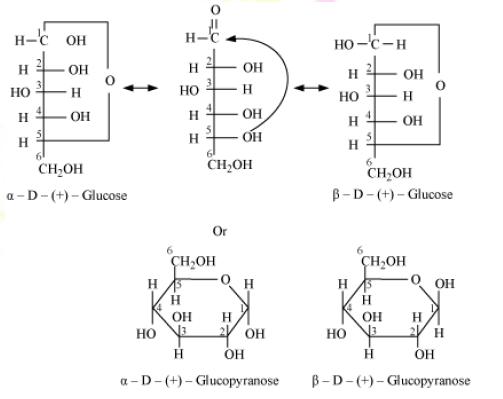

Q17. Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Solution

Q18. Which α-amino acid can cross link peptide chains?
Solution
Cysteine can cross link peptide chains through disulphide bonds.
Q19. (a) What changes occur in the nature of egg proteins on boiling?(b) Name the type of bonding which stabilizes α-helix structure in proteins.
Solution
(a) On boiling, proteins of egg gets denatured. Thus due to coagulation, water gets absorbed.
(b) Regular folding of the backbone of the polypeptide chain due to hydrogen bonding between  and –NH– groups of the peptide bond stabilizes α-helix structure in proteins.
and –NH– groups of the peptide bond stabilizes α-helix structure in proteins.
Comments
Post a Comment