Q1. An organic compound A, C2H5O2N gives on reduction another compound B C2H7N. What are A and B?
Solution
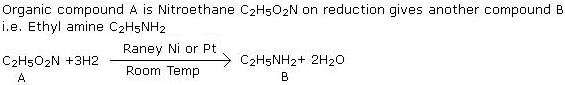
Q2. What happens when amines are treated with water?
Solution
Amines when treated with water it forms alkyl or aryl ammonium hydroxides which ionize to furnish hydroxyl ions.RNH2 + H2O  RNH3OH
RNH3OH  RNH3+ + OH-
RNH3+ + OH-
Q3. Write a note on Gattermann reaction.
Solution
In Gattermann reaction, Chlorine or Bromine can be introduced to the aromatic ring by treating Diazonium salt solution with halogen acids in presence of Copper. 

Q4. Convert benzonitrile to benzaldehyde.
Solution
Q5. Accomplish the following conversions:(i) Nitrobenzene to benzoic acid(ii) Benzene to m-bromophenol
Solution
Q6. Convert Aniline to Phenol.
Solution
Q7. What is a coupling reaction? Give an example.
Solution
Reactions involving retention of Diazo group are coupling reaction. Arenediazonium salts reacts with highly reactive aromatic compounds such as phenols and amines to form brightly coloured azo compounds, Ar-N=N-Ar. The azo products have extended conjugate system having both the aromatic rings joined by N=N bonds. Mostly such compounds are used in the manufacture of dyes, as this compounds are coloured in nature. For example benzene diazonium chloride reacts with phenol to give an orange dye i.e. p-Hydroxyazobenzene. 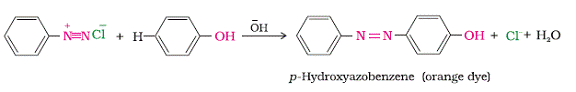

Q8. Write a short note on Hofmann Bromamide reaction?
Solution
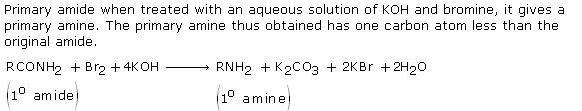
Q9. Write a short note on Schotten-Baumann Reaction?
Solution

Q10. An organic compound A contains C=32%, H=7% and N=20%. On reduction it gives a primary amine B which gives ethyl alcohol and nitrous acid. B gives an offensive odour on warming with CHCl3 and KOH and forms compound C which on reduction forms ethyl methyl amine. Give structures of A, B and C and explain.
Solution
Methyl cyanide on reduction with LiAlH4 yields ethyl amine(A).
Ethyl amine(A) on heating with chloroform and ethanolic potassium hydroxide form ethyl cyanide(C) which is a foul smelling substance.


Q11. How will you convert Methyl halide to Ethylamine?
Solution
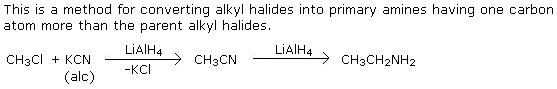
Q12. How can we get primary amines from alkyl cyanides?
Solution
The carbon-nitrogen triple bond of nitriles can be completely reduced to yield primary amines. The reduction is done using H2 in presence of Ni or Pt as catalyst or using LiAlH4.

Q13. What happens when Ethyl amine is treated with Chloroform and Alcoholic potash?
Solution

Q14. Why Cyanides are soluble in water but the corresponding Isocyanides not?
Solution

Q15. What happens when Benzene diazoniumchloride is boiled with water?
Solution
Benzene diazonium chloride is hydrolysed to phenol when heated with water. 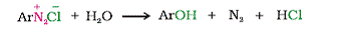
Q16. What product is formed when Aniline is first diazotized and then reacted with Phenol in the alkaline medium?
Solution
p-Hydroxyazobenzene is formed when Aniline is first diazotized and then reacted with Phenol in the alkaline medium.
Q17. What is ammonolysis?
Solution
The process of cleavage of the C-X bond by ammonia molecule is known as ammonolysis.
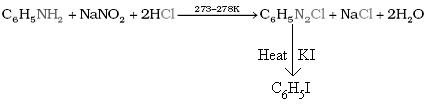
Q18. Convert Aniline to Iodobenzene.
Solution
Comments
Post a Comment