Q1. Complete the following reactions:
(i) (ii)
(ii) 
(iii) CH3CH2Br + NaI
(i)
 (ii)
(ii) 
(iii) CH3CH2Br + NaI
Solution
(i)

 + SO2 + HCl
(ii)
+ SO2 + HCl
(ii)

 (iii) CH3CH2Br + NaI
(iii) CH3CH2Br + NaI  CH3CH2I + NaBr
CH3CH2I + NaBr

 + SO2 + HCl
(ii)
+ SO2 + HCl
(ii)

 (iii) CH3CH2Br + NaI
(iii) CH3CH2Br + NaI
Q2. Why is sulphuric acid not used during the reaction of alcohols with KI?
Solution
H2SO4 converts KI to corresponding HI and then oxidizes it to I2. Hence it cannot be used during the reaction of alcohols with KI.
Reaction:
KI(s) + H2SO4(l) → KHSO4(s) + HI(g) 8H+ + 8I- + H2SO4 (l) → H2S (g) + 4H2O (l) + 4I2(s)
Q3. What are freons? Give one example.
Solution
The chlorofluorocarbon compounds of methane and ethane are collectively known as freons.
Example: Freon-12( CCl2F2)
Q4. What are enantiomers? Give an example.
Solution
The stereoisomers related to each other as nonsuperimposable mirror images are called enantiomers.
For example:
2-chloro butane (CH3CH2 C*H (Cl) CH3).
Here the carbon atom marked is the chiral centre and posseses four different groups.
Q5. Give the IUPAC name of the compound CH3CH (Cl) CH (Br) CH3.
Solution
2-bromo-3-chloro-Butane
Q6. Define racemisation.
Solution
A mixture containing two enantiomers in equal proportions is known as racemic mixture and the process of conversion of enantiomers into a racemic mixture is known as racemisation.
Q7. Give the three uses of each of the following: (i) Dichloromethane (ii) Tetrachloromethane
Solution
Dichloromethane: It can be used (i) As solvent (ii) As paint remover (iii) As aerosol propellant Tetrachloro methane: It can be used (i) As a solvent (ii) As a fire extinguisher (iii) As a degreasing agent
Q8. Which alkyl halides show colour on exposure to light?
Solution
Alkyl Bromide and Alkyl iodide.
Q9. Which compound in each of the following pairs will react faster in SN2 reaction with -OH? (i) CH3Br or CH3I (ii) (CH3)3CCl or CH3Cl
Solution
(i) CH3I will react faster than CH3Br because the order of reactivity is as follows: R-I > R-Br > R-Cl > R-F
(ii) CH3Cl will react faster than (CH3)3CCl because the order of reactivity is as follows in SN2 reaction.
Primary halide > Secondary halide > Tertiary halide
Q10. What is DDT and why its use is banned in United States?
Solution
DDT is p, p'-Dichlorodiphenyltrichloroethane and is used as insecticide.
It is banned for the following reasons:(i) Its high toxicity towards fish.(ii) DDT is not metabolized very rapidly by animals instead it gets deposited and stored in the fatty tissues.
Q11. Why the use of chloroform as anesthetic is decreasing?
Solution
Inhaling chloroform vapors depresses the central nervous system and its chronic exposure may cause damage to the liver and kidneys due to metabolism of chloroform to phosgene gas. Hence the use of chloroform as anesthetic is decreasing.
Q12. Why chloroform is kept closed in dark colored bottles?
Solution
Chloroform is oxidized slowly by air in the presence of light to an extremely poisonous gas, carbonyl chloride, also known as Phosgene
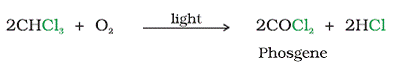 Hence it is kept closed in dark colored bottles.
Hence it is kept closed in dark colored bottles.
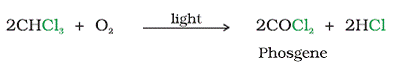 Hence it is kept closed in dark colored bottles.
Hence it is kept closed in dark colored bottles.
Q13. Give reasons for the following:
i) Haloalkanes react with KCN to form alkyl cyanides as main product while AgCN forms isocyanide as the chief products.
ii) Alcohols do not react with NaBr, but when H2SO4 is added, they form alkyl bromides.
Solution
i) KCN is predominantly ionic and provides cyanide ions in solution. Although both carbon and nitrogen atoms of CN can donate electron pairs but the attack takes place mainly through carbon atom and not through nitrogen atom because C-C bond is more stable than C-N bond. However AgCN is mainly covalent in nature and nitrogen is free to donate electron pair forming isocyanide as the main product.
ii) Br- is a weak base. So it cannot displace the strong base OH- when H2SO4 is added, it leads to protonation of alcohol, as a result of which water molecule is formed. Since water molecule is a very weak base it is easily replaced by Br-.
Q14. A primary alkyl halide (A) C4H9 reacts with alcoholic KOH to give a compound (B). The compound (B) reacts with HBr to give the compound C which is an isomer of A. When A reacts with sodium metal it give a compound (D) whose molecular formula is C8H18. The compound D is different from the compound formed when n-butyl bromide reacts with sodium. Give the structural formula of A and write all the equations involved in the reaction.
Solution
For C4H9Br, two primary halides are possible CH3CH2CH2CH2Br (n-butyl bromide) and (CH3)2CHCH2Br (iso-butyl bromide). Since A is not normal –butyl bromide, it must be iso-butyl bromide.
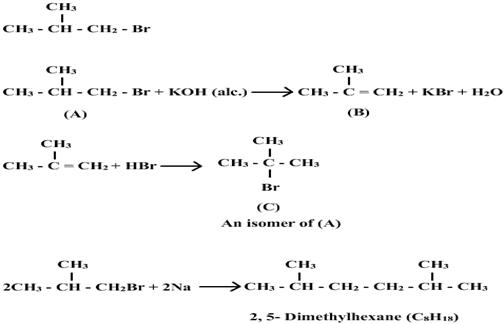

Q15. Write the structure of the major organic product in each of the following reactions:
(i) CH3CH2CH2OH + SOCl2  (ii) CH3CH2CH = CH2 + HBr
(ii) CH3CH2CH = CH2 + HBr  (iii) CH3CH = C (CH3)2 + HBr
(iii) CH3CH = C (CH3)2 + HBr 
Solution
(i) CH3CH2CH2Cl
(ii) CH3CH2CH2CH2Br
(iii) CH3CH2 CBr(CH3)2
Q16. Give reasons for the following: (i) Grignard reagent should be prepared under anhydrous conditions. (ii) Neo-pentyl bromide undergoes nucleophilic substituting reactions very slowly. (iii) p-Methoxybenzyl bromide reacts faster than p-nitrobenzyl bromide with ethanol to form an ether product.
Solution
(i) Grignard reagent reacts with water and gets decomposed, so it is produced in anhydrous conditions. (ii) Neo-pentyl bromide being a primary halide reacts slowly through SN1, and being a sterically hindered halide reacts slowly even through SN2 mechanism. (iii) Methoxy (-OCH3) group being an electron releasing group stabilizes the intermediate carbocation. On the other hand nitro group (-NO2) is an electron withdrawing group and hence it destabalises the intermediate carbocation. Thus p-methoxy benzyl bromide reacts faster than p-nitrobenzyl bromide because the reaction proceeds via more stable intermediate carbocation.
(ii) Neo-pentyl bromide being a primary halide reacts slowly through SN1, and being a sterically hindered halide reacts slowly even through SN2 mechanism. (iii) Methoxy (-OCH3) group being an electron releasing group stabilizes the intermediate carbocation. On the other hand nitro group (-NO2) is an electron withdrawing group and hence it destabalises the intermediate carbocation. Thus p-methoxy benzyl bromide reacts faster than p-nitrobenzyl bromide because the reaction proceeds via more stable intermediate carbocation.
Q17. Write the equations for the preparation of 1-iodobutane from: (i) 1-butanol (ii) 1-chlorobutane (iii) But-1-ene
Solution
(i) CH3CH2CH2CH2OH  CH3CH2CH2CH2I
1-butanol 1-iodobutane
(ii) CH3CH2CH2CH2Cl + NaI
CH3CH2CH2CH2I
1-butanol 1-iodobutane
(ii) CH3CH2CH2CH2Cl + NaI  CH3CH2CH2CH2I + NaCl
1-chlorobutane 1-iodobutane
(iii) CH3CH2CH = CH2 + HI
CH3CH2CH2CH2I + NaCl
1-chlorobutane 1-iodobutane
(iii) CH3CH2CH = CH2 + HI  CH3CH2CH2CH2I
But-1-ene. 1-iodobutane
CH3CH2CH2CH2I
But-1-ene. 1-iodobutane
Comments
Post a Comment