Q1. Compare the most preferred packing of solids to the least preferred packing of solids.
Solution
The most preferred packing is face centered cubic by solids and the least preferred packing is simple cube. The packing efficiency is maximum in face centered cube so it is very stable. If we compare the values obtained for the two unit cells, we see that "packing efficiency" increases in the face-centered cubic 74%> simple cubic 52%.
Q2. Why ancient glass becomes milky?
Solution
Amorphous solids become crystalline on ageing e.g. ancient glass appears milky due to
Q3. The packing efficiency of the two-dimensional square unit cell shown below is
Q3. The packing efficiency of the two-dimensional square unit cell shown below is
Solution
The packing efficiency of the two-dimensional square unit cell is 78.54%.
Q4. The number of atoms per unit cell in a face centred cubic arrangement is:
Solution
For an fcc unit cell,
Number of atoms per unit cell = (8 × 1/8) + ( 6 × 1/2) = 4
Q5. What is the co-ordination number of atoms in BCC, HCP, CCP and simple lattices?
Solution
The co-ordination number of atoms in BCC lattice is 8, in HCP lattice 12, in CCP lattice 12 and in simple lattice it is 6.
Q6. The fraction of the total volume occupied by the atoms present in a simple cube is
Solution
The fraction of the total volume occupied by the atoms present in a simple cube is  .
.
 .
.
Q7. If 300 atoms form close packing how many tetrahedral void and octahedral void will be formed
Solution
If n number of atoms form close packing than 2n tetrahedral voids & n Octahedral voids are formed.For 300 atoms forming close packing 600 tetrahedral void and 300 octahedral void will be formed.
Q8. How many atoms are there in the given cubic unit cell?

Solution
In a simple cubic unit cell total number of atoms will be present at the 8 corners. It comes out to be equal to 8 x 1/8= 1 atom.
Q9. Under what conditions will sodium chloride conduct electricity.
Solution
Sodium chloride is ionic solids made of sodium and chloride ions bonded to each other by ionic bond. In solid state the high binding energy keeps the ion in fixed sites permitting only rotational and vibrational movement. In molten state the movement of ions is not restricted so in electrical field the ions move towards the opposite field.
Q10. The number of atoms per unit cell in a body centred cubic arrangement is:
Solution
For a bcc unit cell, number of atoms per unit cell = (8 × 1/8) + 1 = 2
Q11. A compound is formed by two elements X and Y. Atoms of the element Y (as anions) make ccp and those of element X (as cations) occupy all the octahedral voids the formula for the compound is:
Solution
The ccp lattice is formed by the element Y. The number of octahedral voids generated would be equal to the no of atoms of Y present in it. Since all the octahedral voids are occupied by the atoms of X, the number would also be equal to that of the element Y. Therefore the formula of the compound is XY.
Q12. Three elements X, Y and Z crystallise in a cubic solid with X at corners, Y at body centre and Z at the face centres of the cube, then the formula of the solid is
Solution
There is one atom of X, one atom of Y and 3 atoms of Z.
Q13. With example explain F-center.
Solution
F-centers is the anionic site occupied by the electron ( e– becomes part of the lattice). These electrons are color centers, often referred to as F-centers (from the German word farben meaning color).
For example, when NaCl is heated in an atmosphere of Na vapour, the excess of Na atoms deposit on the surface of NaCl crystal. Cl− ions then diffuse to the surface where they combine with the Na atoms which becomes ionized by losing electrons. These electrons diffuse back into the crystal and occupy the vacant sites created by the Cl− ions. These electrons absorb some energy of the white light. giving yellow colour to NaCl. Similarly, excess of Li in LiCl makes it pink and excess of K in KCl makes it violet.
Q14. Give two examples each of (a) Molecular (b) Ionic (c) Covalent (d) Metallic.
Solution
Types of solids
Molecular e.g. ice, sugar
Metallic e.g.
Silver,gold
Covalent e.g.sand,
diamond)
Ionic e.g.
Common salt NaCl and potassium chloride.
Q15. An element A has a ccp arrangement of atoms. The radius of A is equal to 143.2pm. Calculate the lattice parameter of the unit cell and the density of solid Al if its atomic weight = 30u.
Solution
Because A is ccp we have an fcc unit cell. Cell contents: 4 atoms/cell [8 at corners (each 1/8), 6 in faces (each 1/2)]
Lattice parameter: atoms in contact along face diagonal, therefore 4rAl = a(2)1/2
a = 4(1.432Å)/(2)1/2 = 4.050Å.
Density = Mass/Volume = Mass per unit cell/Volume per unit cell g/cm3
Mass of unit cell = mass of 4 Al atoms = (30g/mol)(1mol/6.022x1023atoms)(4 atoms/unit cell) = 1.992 x 10-22 g/unit cell
Volume unit cell = a3 = (4.05x10-8cm)3 = 66.43x10-24 cm3/unit cell
Density= {1.992x10-22g/unit cell}/{66.43x10-24 cm3/unit cell} = 2.998 g/cm3
Q16. If formula of the compoud is A2B3 A forms the close packing how many octahedral voids are occupied.
Solution
In this compound A forms the close packing and B occupies the 2/3 the octahedral void. Ratio will be n A: 2/3 n B.
Q17. In which of the following solids, ions of opposite charges are held together by strong electrostatic forces of attraction?
Solution
Ionic solids contain oppositely charged ions held together by electrostatic forces of attraction also called as ionic bonds.
Q18. An element has a body centred cubic (bbc) structure with a cell edge of 288 pm. The density of the element is 7.2 g/cm3. How many atoms are there in 208 g of the element?
Solution
Volume = 288 pm = (288 x 10-10 cms)3 = 2.39 x 10 -23 cm 3
Volume of 208 g of the element = mass/density = 208 g / 7.2 g cm-3 = 28.88 cm3
Number of unit cells in this volume = 28.88 cm3/2.39 x 10 -23 cm3/unit cell = 12.08 x1023 unit cells.
Since each bcc cubic unit cell contains 2 atoms, therefore, the total number of atoms in 208g = 2 atoms /unit cell x 12.08 1023 unit cells = 24.16 x 1023atoms
Q19. Element crystallizes as BCC. How many atom will be present in the unit cell of the element.
Solution
The body-centered cubic unit cell has atoms at each of the eight corners of a cube plus one atom in the center of the cube. Corners will always be 1/8 and 1 in the center.
8 corners x 1/8 = 1 and Body center = 1x1 therefore total 2 atoms per unit cell. So the unit cell of the element will have 2 atoms.
Q20. With an example explain glass is a super cooled liquid or a pseudo solid.
Solution
An amorphous substance, such as glass, tends to be isotropic. This difference may make it possible to distinguish between a glass and a crystal. Amorphous materials, like window glass, have no long-range order at all, so they have no translational symmetry. The structure of an amorphous solid (and indeed a liquid) is not truly random - the distances between atoms in the structure are well defined and similar to those in the crystal. This is why liquids and crystals have similar densities - both have short-range order that fixes the distances between atoms, but only crystals have long-range order. Amorphous materials like glass have no 'distinct' crystal directions, so anisotropic properties are generally not observed. For example Glass panes fixed to old building are thicker at the bottom than the top because amorphous solids do not melt at a sharp melting point. They soften over a range of temperature and can be molded and blown. Like liquids amorphous solids have a tendency to flow very slowly and the movement is not noticeable. Therefore they are called super cooled liquids or pseudo solids.
Q21. In a solid, oxide ions are arranged in CCP, cation A occupy one- sixth of the tetrahedral voids and cations B occupy one third of the octahedral voids. What is the formula of the compound?
Solution
In CCP, there would be 2 tetrahedral voids and 1 octahedral void. One-third octahedral voids are occupied by B while one-sixth tetrahedral voids are occupied by A. Thus, formula of the compound may be derived as
A1∕3B2∕6O or AB2O6 or ABO3.
Q22. Give coordination number of octahedral void.
Solution
Coordination number is 6. The space unoccupied between 4 spheres in one plane & 2spheres in the axis and space occupied is 74%.
Q23. Why is LiCl in excess of Li pink in color.
Solution
LiCl in vapor of Li an electron occupies anionic site called F-center and the crystal imparts pink color. F centers is the anionic site occupied by the electron ( e– becomes part of the lattice) These electrons are color centers, often referred to as F-centers (from the German word farben meaning color).
Q24. In AB2O4, the oxide ions are placed in cubic close packed lattice, cation A is present in tetrahedral voids and cations B are present in octahedral voids. What is the percentage of tetrahedral and octahedral voids occupied by A and B respectively?
Solution
In CCP lattice of oxide ions, there would be two tetrahedral voids and one octahedral void for each oxide ion. Therefore for four oxide ions, there would be 8 tetrahedral and 4 octahedral voids. Out of 8 tetrahedral voids, 1 is occupied by
A and out of 4 octahedral voids 2 are occupied by B
% of tetrahedral voids occupied by A =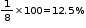 % of octahedral voids occupied by B =
% of octahedral voids occupied by B =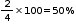
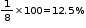 % of octahedral voids occupied by B =
% of octahedral voids occupied by B =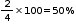
Q25. Explain dislocation.
Solution
Dislocations means linear defects which occur when a crystal structure contains misaligned planes of atoms, atoms are ‘dislocated’ from their position in the lattice.
Q26. How much space is filled in cubic close packing?
Solution
Cubic close packing is highly efficient and 74% space in the crystal is filled.
Q27. Calculate the density for nickel if the unit cell edge length for nickel given is 0.3524 nm and atomic mass is 58.69 g /mol. It crystallizes as a simple cube.
Solution
The volume (V) of the unit cell is equal to the cell-edge length (a) cubed.
a =.3524nm =.352 x 10-7cm
V = a3 = (0.3524 x 10-7cm )3 =4.376 x10-23cm3
The mass of a nickel atom can be calculated from the atomic weight of
this metal and Avogadro's number. 58.69g/mol / 6.022 x 1023
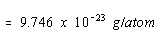 The density of nickel, =9.746 x 1023 /4.376 x 1023 = 2.23 g/cm3
The density of nickel, =9.746 x 1023 /4.376 x 1023 = 2.23 g/cm3
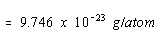 The density of nickel, =9.746 x 1023 /4.376 x 1023 = 2.23 g/cm3
The density of nickel, =9.746 x 1023 /4.376 x 1023 = 2.23 g/cm3
Q28. A FCC lattice cube is formed by atoms A and B. If atom A is present at the corner of the cube and the atom B at the faces of the cube. Find out the formula of the compound?
Solution
Contribution of atom A at eight corners of the cube = 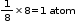 Contribution of atom at each face =
Contribution of atom at each face = 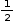 atom
Contribution of atom B at six faces of the cube =
atom
Contribution of atom B at six faces of the cube = 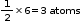 Formula of the compound = AB3
Formula of the compound = AB3
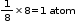 Contribution of atom at each face =
Contribution of atom at each face = 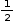 atom
Contribution of atom B at six faces of the cube =
atom
Contribution of atom B at six faces of the cube = 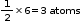 Formula of the compound = AB3
Formula of the compound = AB3
Q29. Explain Non-stoichiometric defects in solids.
Solution
These defects disturb the stoichiometry of the compound and are of two types: Metal excess defect due to anionic vacancy or extra cations & Metal deficiency defect.
Metal excess defect: ZnO is white and lose oxygen on heating, and the excess Zn metal atoms in the sample are ready to give electrons. Electrons in defect region only absorb light and it becomes yellow. Replacement of Al3+ for Si4+ in quartz gives rise to the color of smoky quartz.
Metal deficiency defect. Sometimes the crystal is not formed as per the stoichiometric proportions e.g. FeO is more typically is found as FexO where x~0.95-0.99.
Charge neutrality must be maintained so some (Fe2+)are missing and the loss is made up by (Fe3+) in FeO crystal.
Q30. What is the difference between Schottky and Frenkel defects.
Solution
A Frenkel defect is form of interstitial defect when an atom or ion leaves its place in the lattice (leaving a vacancy), and lodges nearby in the crystal (becoming an interstitial). The number of interstitials formed will equal the number of vacancies formed and the density remains the same.
Schottky defects: is a vacancy defect , it decreases the density and equal numbers of cations and anions are missing from the lattice.
Q31. Which type of crystals show anisotropy.Describe anisotropy.
Solution
In crystalline solids you can see there is long range order. Arrangement of particles in crystalline solids is different along different directions so the value of properties like refractive index or electrical resistance vary in different direction such solids are anisotropic. Crystals will melt at a fixed temperature called melting point. Crystalline solids when melted and cooled rapidly form amorphous solid.
Q32. Name and illustrate the parameter that characterise a unit cell in a crystal?
Solution
Characteristics of Unit cells based on distance between particles in the x- axis, y- axis & z- axis depicted by a, b & c and the angle between the edges in the three axis given by α, β & γ as shown in the figure below.


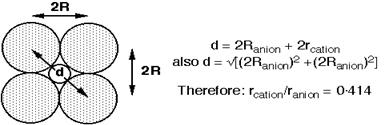
Q33. If the radius of the cation is r present in octahedral void and radius of the anion in close- packing is R, derive relation between r and R.
Solution
Relation between r and R.

Q34. If atom A forms the close packing B occupies 1/4 the octahedral void and C occupies the 1/4 the tetrahedral void, find the formula of the compound.
Solution
Step 1--- If n number of A forms the close packing 2n tetrahedral voids will be formed and n octahedral void will be formed.
Step 2---- If 1/4 the Tetrahedral voids and 1/4 the octahedral voids are occupied then the ratio will be nA : n x 1/4 B : 2n x 1/4 C = A : 1/4 B : 1/2 C, formulas are whole number so multiply by 4
Step 3--- The formula of the compound is A4BC2
Q35. How many octahedral voids and tetrahedral voids are formed in one cube.
Solution
Octahedral voids are formed by 6 spheres and so there will be 4 octahedral voids in a cube and tetrahedral voids are formed by 4 spheres so there are 8 tetrahedral voids in a cube.



Comments
Post a Comment